.
Similarly, it is asked, what is the difference between an electrochemical cell and a battery?
The difference between a cell and a battery is that a cell is a single unit that converts chemical energy into electrical energy, and a battery is a collection of cells. Aggregating cells into a battery increases the electrical voltage they produce. Batteries are classified by electrolyte type.
Also Know, are batteries chemical energy? A battery is a device that stores chemical energy and converts it to electrical energy. The chemical reactions in a battery involve the flow of electrons from one material (electrode) to another, through an external circuit.
Beside this, are batteries galvanic cells?
A battery is a package of one or more galvanic cells used for the production and storage of electric energy by chemical means. A galvanic cell consists of at least two half cells, a reduction cell and an oxidation cell. Chemical reactions in the two half cells provide the energy for the galvanic cell operations.
Do voltaic cells need batteries?
A voltaic cell produces electricity as a redox reaction occurs. The voltage of a voltaic cell can be determined by the reduction potentials of the half reactions. Voltaic cells are fashioned into batteries, which are a convenient source of electricity.
Related Question AnswersWhat are the 2 types of electrochemical cells?
Two Types of Cell There are two fundamental types of electrochemical cell: galvanic and electrolytic. Galvanic cells convert chemical potential energy into electrical energy. The energy conversion is achieved by spontaneous (ΔG < 0) redox reactions producing a flow of electrons.What is a cell of battery?
Cell. A cell refers to a single anode and cathode separated by electrolyte used to produce a voltage and current. A battery can be made up of one or more cells. A single AA battery, for example, is one cell. Car batteries contain six cells at 2.1 V each.Why is a battery called a cell?
A chemical reaction takes place in the battery when it is discharged that produces a flow of electrons out one plate on the negative side and into another plate on the positive side. Actually a single unit of a battery is a cell. A battery is called a "battery", because it is a "battery" of cells together.What are the two main components of an electrochemical cell?
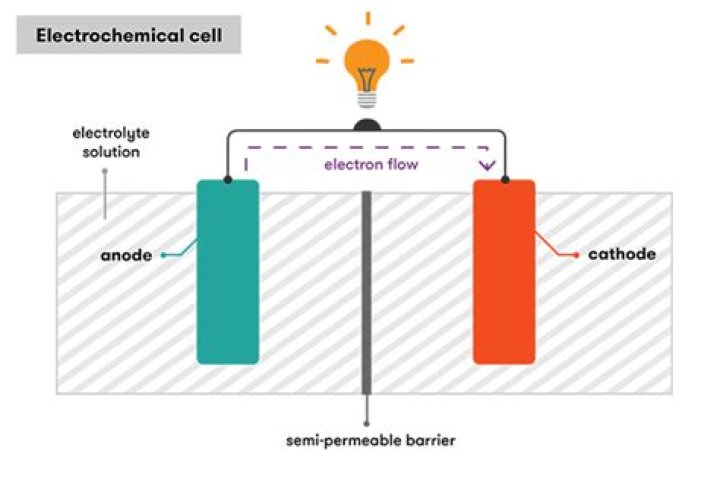
Electrochemical cells have two conductive electrodes, called the anode and the cathode. The anode is defined as the electrode where oxidation occurs. The cathode is the electrode where reduction takes place.Is a cell a battery?
The difference between cell and battery is that a cell is a single unit that converts chemical energy into electrical energy, and a battery is a collection of cells.How do you represent a cell?
Cell notation. Cell notation or line notation in chemistry is a shorthand way of expressing a certain reaction in an electrochemical cell. The cell anode and cathode (half-cells) are separated by two bars or slashes representing a salt bridge, with the anode on the left and cathode on the right.What do you mean by anode?
An anode is the electrode in a polarized electrical device through which current flows in from an outside circuit. Cathodes get their name from cations (negatively charged ions) and anodes from anions (positively charged ions). In a device that consumes electricity, the anode is the charged positive electrode.What is a cell in a circuit?
An electrical cell is an "electrical power supply" - internally it converts stored chemical energy into electrical potential energy, allowing a current to flow from the positive terminal round to the negative one via an external circuit (this is called conventional current, which is chosen to go from + to -).How can I make a battery?
Method 1 Making a Soda-powered Battery- Gather your materials.
- Fill the plastic cup roughly 3/4 full with soda.
- Make sure the soda can is completely empty.
- Cut a strip of aluminum from the soda can.
- Sand the aluminum strip (optional).
- Place the strips into the solution.
- Attach lead wires to metal strips.