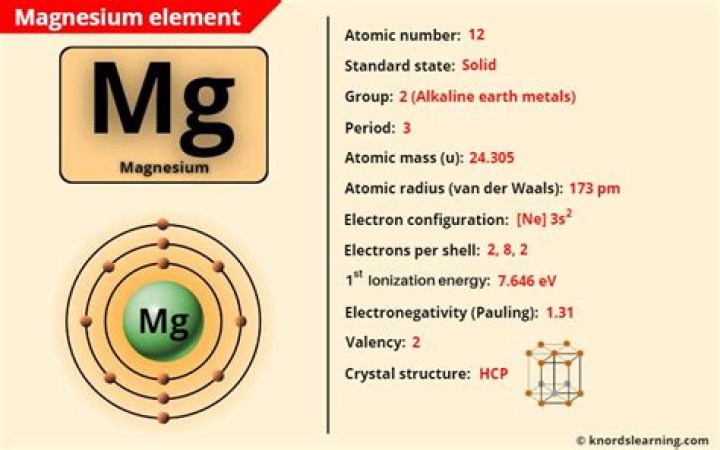
The structures of the elements change as you go across the period. The first three are metallic, silicon is giant covalent, and the rest are simple molecules. Sodium, magnesium and aluminium all have metallic structures. In magnesium, both of its outer electrons are involved, and in aluminium all three..
Likewise, people ask, why is sodium in Period 3 of the periodic table?
Argon, chlorine, sulfur and phosphorus are non-metals. On the left hand side of period 3 we find the elements sodium, magnesium and aluminium. The more electrons a metallic element can contribute to the metallic bond the more mobile electrons there will be and the stronger the metallic bond will be!
what metal in period 3 is more reactive than magnesium? These metals are less reactive than the neighboring alkali metal. Magnesium is less active than sodium; calcium is less active than potassium; and so on. These metals become more active as we go down the column. Magnesium is more active than beryllium; calcium is more active than magnesium; and so on.
Also to know, what is Period 3 on the periodic table?
The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block.
Why does conductivity decrease across Period 3?
Explanation of this trend They have metallic bonding, in which the nuclei of metal atoms are attracted to delocalised electrons. Going from sodium to aluminium: there are more electrons that can move and carry charge through the structure … the electrical conductivity increases.
Related Question Answers
What is the largest atom in period 3?
(B) Trends in the Atomic Radius of Elements in Period 3
| Element | Na | Cl |
| Energy Level being filled (Valence Shell) | third (M) | third (M) |
| Nuclear Charge (charge on all protons) | 11+ | 17+ |
| Atomic Radius (pm) | 154 | 99 |
| General Trend | (largest) | → |
Which is the most reactive metal?
cesium
What are the periodic trends in electronegativity?
Electronegativity increases as you move from left to right across a period on the periodic table. This is because, even though there are the same number of energy levels, there are more positive protons in the nucleus, creating a stronger pull on the negative electrons in the outer shell.Is Melting Point A periodic property?
Melting points and boiling points show periodic properties. This means that they vary in a regular way or pattern depending on their position in the Periodic Table.Does Period 3 have 5 valence electrons?
Phosphorous is the 5th element from the left in the third period so it has five valence electrons.Are Group 3 elements metals?
Group 3 is a group of elements in the periodic table. When the group is understood to contain all of the lanthanides, it subsumes the rare-earth metals. Yttrium, and less frequently scandium, are sometimes also counted as rare-earth metals.What is the 3 types of elements?
Classification of the Elements The next thing in our review is to classify the elements into three groups. These three groups are: metals, nonmetals, and inert gases. Let's look at where these groups are located on the periodic table and correlate them with the ability to lose and gain electrons.What is the most metallic element in period 3?
The element in Period 3 with the most metallic character is (A) sodium; (B) aluminum; (C) silicon; (D) phosphorus.What is Group 14 on the periodic table called?
The carbon group is a periodic table group consisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). In modern IUPAC notation, it is called Group 14. In the field of semiconductor physics, it is still universally called Group IV.What are the 7 periods of the periodic table?
The 7th period of the periodic table now has four new elements: element 113 (temporarily named as Ununtrium, or Uut), element 115 (Ununpentium, or Uup), element 117 (Ununseptium, or Uus), and element 118 (Ununoctium, or Uuo), says a group of experts from the International Union of Pure and Applied Chemistry (IUPAC) andWho discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.Why is Aluminium in period 3?
In the whole of period 3, the outer electrons are in 3-level orbitals. That increases ionisation energies still more as you go across the period. The fall at aluminium. You might expect the aluminium value to be more than the magnesium value because of the extra proton.Is calcium more reactive than magnesium?
Answer and Explanation: Calcium is more reactive than magnesium because the calcium atom is larger than the magnesium atom. Because of its larger size, calcium has one moreIs calcium more reactive than potassium?
Calcium has two valence electrons while Potassium only has one. For the people who just want an answer for their homework; Potassium is more reactive because it's single valence electron requires less energy to be transferred in a reaction than for both of Calcium's valence electrons to transfer in a reaction.What is the name of the 4th halogen?
Halogens on the Right In the second column from the right side of the periodic table, you will find Group Seventeen (Group XVII). This column is the home of the halogen family of elements. Who is in this family? The elements included are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At).Why does the third period contain 8 elements but not 18?
The electron capacity of third shell is 18. So the third period should actually contain 18 elements. But it contains only 8 elements due to the law of electron octet. This is because of the fact that electrons are filled in these shells in the increasing order of electrons.What element is in Group 2 Period 3?
Argon
How many transition metals are there in period 3?
The 38 elements in groups 3 through 12 of the periodic table are called "transition metals". As with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat.