.
Also, what is reduction in organic chemistry?
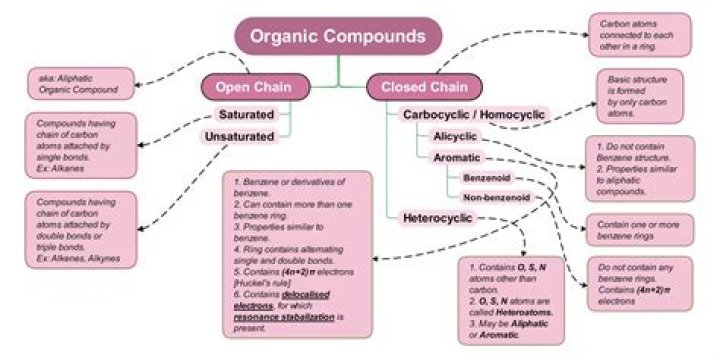
In reference to organic molecules, oxidation is a process by which a carbon atom gains bonds to more electronegative elements, most commonly oxygen. Reduction is a process by which a carbon atom gains bonds to less electronegative elements, most commonly hydrogen.
One may also ask, what is the most reduced molecule? The most reduced form of carbon is CH4, the most oxidized is CO2. Thus the oxidation state of a one-carbon fragment is unambiguous and defined by the number of C-H bonds that have been replaced by C-X bonds, where X = any electronegative element (see periodic table on previous page).
Subsequently, one may also ask, what are reduced compounds?
A reduced compound (one capable of being reduced), therefore, must bear bonds to atoms that are less electronegative than its oxidized form. A reduced compound (one capable of being reduced), therefore, must bear bonds to atoms that are less electronegative than its oxidized form.
Why does the oxidation of organic compounds?
The covalent bonds in organic molecules are higher energy bonds than those in water and carbon dioxide. B. Electrons are being moved from atoms that have a lower affinity for electrons (such as C) to atoms with a higher affinity for electrons (such as O). The oxidation of organic compounds can be used to make ATP.
Related Question AnswersWhat is oxidation VS reduction?
Oxidation vs Reduction Oxidation occurs when a reactant loses electrons during the reaction. Reduction occurs when a reactant gains electrons during the reaction.Which functional group is the most oxidized?
1 Answer- Thus, the CHO group is more highly oxidized than the CH2OH group.
- C-1 in the alcohol group has two H atoms attached, while C-1 in the aldehyde group has one H atom attached.
- Therefore, the CHO group is more highly oxidized than the CH2OH group.
What is reduction reaction with example?
Reduction can be considered as the removal of oxygen, the addition of hydrogen, or the gain of electrons. Removal of oxygen: Metal ores which are oxides are reduced to the metal - this is how iron is made from iron ore. The reducing agent is carbon monoxide.What is oxidation of organic matter?
Aerobic respiration is an oxidation reaction in which carbon in organic matter is oxidized to carbon dioxide with the release of energy. Respiration by microorganisms decomposing organic matter in pond soil consumes oxygen faster than it can penetrate the soil mass, and only the surface layer is aerobic.What do you mean by reduction reaction?
Reduction involves a half-reaction in which a chemical species decreases its oxidation number, usually by gaining electrons. In some reactions, oxidation and reduction may be viewed in terms of oxygen transfer. Here, oxidation is the gain of oxygen, while reduction is the loss of oxygen.How do you know if its oxidized or reduced?
Identifying the Elements Oxidized and Reduced- Assign oxidation numbers to all atoms in the equation.
- Compare oxidation numbers from the reactant side to the product side of the equation.
- The element oxidized is the one whose oxidation number increased.
- The element reduced is the one whose oxidation number decreased.
What is oxidation in chemistry?
Oxidation is the loss of electrons during a reaction by a molecule, atom or ion. Oxidation occurs when the oxidation state of a molecule, atom or ion is increased. The opposite process is called reduction, which occurs when there is a gain of electrons or the oxidation state of an atom, molecule, or ion decreases.What compounds are mainly used as oxidizing agents in organic chemistry?
Common oxidizing agents are oxygen, hydrogen peroxide and the halogens. In one sense, an oxidizing agent is a chemical species that undergoes a chemical reaction in which it gains one or more electrons.Is Oxygen an oxidizer?
No oxygen is not always an oxidizer or oxidising agent. For it to be so it needs to be reduced, I.e. It needs to gain electrons. For the most part oxygen almost always does this however, if it gets bonded to a much more electro negative atom such as fluorine then oxygen becomes oxidised and the fluorine reduced.Why is it called oxidation?
In the early days of chemistry, oxidation was defined as a gain of oxygen atoms. the Mg was said to be oxidized because it gained an oxygen atom. Eventually, chemists realized that the reaction involved a transfer of electrons from Mg to O .Is losing a hydrogen oxidation or reduction?
The original view of oxidation and reduction is that of adding or removing oxygen . An alternative approach is to describe oxidation as the loss of hydrogen and reduction as the gaining of hydrogen. The CO is reduced because it gains hydrogen, and the hydrogen is oxidized by its association with the oxygen.Is oxidation a rust?
A classic example of a redox reaction is rusting. When rusting happens, oxygen steals electrons from iron. Oxygen gets reduced while iron gets oxidized. The result is a compound called iron oxide, or rust.What is a good oxidizing agent?
This means that they easily attract electrons. As you can see in the periodic table of elements, the halogens that are good oxidizing agents are fluorine, chlorine, bromine and iodine, with fluorine being the strongest oxidizing agent among the four, followed by chlorine, bromine and iodine.Is respiration a redox reaction?
In summary, cellular respiration is the biological process in which organic compounds are converted into energy. A redox reaction is shorthand for an oxidation-reduction reaction and is a chemical reaction in which one molecule loses electrons while another molecule gains electrons. C6H12O6 + 6O2 -> 6CO2 + 6H2O.What is it called when a substance loses hydrogen?
Oxidation is a process where a substance. Loses one or more electrons. Gains an oxygen atom or Electronegative atoms. Loses a hydrogen atom or Electropositive atoms.What is the most common molecule in the body?
The Most Important Molecules in Your Body- Water. Water is an essential molecule for life.
- Oxygen. About 20% of air consists of oxygen.
- DNA. DNA codes for all the proteins in the body, not just for new cells.
- Hemoglobin. Hemoglobin is a macromolecule that transports oxygen in red blood cells.
- ATP.
- Pepsin.
- Cholesterol.
How do you get the oxidation number?
1 Answer- The oxidation number of a free element is always 0.
- The oxidation number of a monatomic ion equals the charge of the ion.
- The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements.
- The oxidation number of O in compounds is usually -2, but it is -1 in peroxides.
How do you determine oxidation level?
To determine the oxidation level of an atom within a molecule, separate the atom from its bonding partner(s), assigning all bonding electrons to the more electronegative of the bonded atoms. Then compare the number of electrons that "belong" to each atom to the atomic number of that atom.How do you know which carbon is most oxidized?
To calculate the oxidation state for carbon, use the following guidelines:- In a C-H bond, the H is treated as if it has an oxidation state of +1.
- For carbon bonded to a more electronegative non-metal X, such as nitrogen, oxygen, sulfur or the halogens, each C-X bond will increase the oxidation state of the carbon by 1.