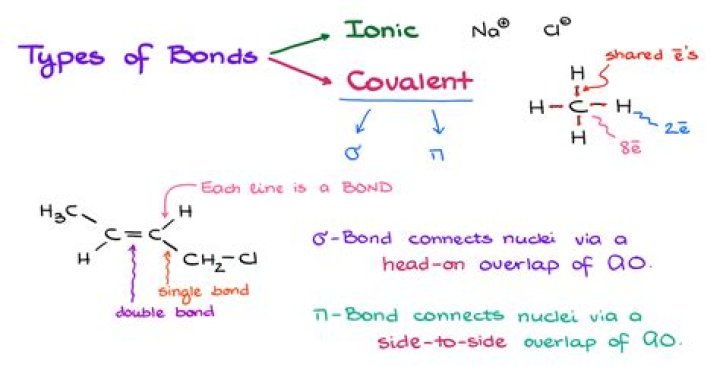
.
Keeping this in consideration, what is the molecular formula for lithium?
Lithium
| PubChem CID: | 3028194 |
|---|---|
| Chemical Safety: | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula: | Li |
| Synonyms: | Lithium 7439-93-2 litio Lithium, metallic Lithium, elemental More |
| Molecular Weight: | 7 g/mol |
Similarly, is AlI3 aqueous? Aluminum iodide is an ionic compound with the formula AlI3. Aluminum iodide is soluble in water, ethanol, diethyl ether, carbon disulfide, sulfur dioxide and pyridine.
Moreover, is AlI3 polar or nonpolar?
The dipole moments of each of the Al-Cl bond is directed at 120 degree angles to each other in a plane, and is therefore cancelled out. Therefore it is a non-polar molecule.
Is Aluminium iodide ionic or covalent?
Aluminium iodide is covalent because the electron pair is easily dragged away from the iodide ion. On the other hand, aluminium fluoride is ionic because the aluminium ion can't polarise the small fluoride ion sufficiently to form a covalent bond.
Related Question AnswersIs there enough lithium?
With the advent of the electric vehicle, the demand could skyrocket but for now the world has enough proven lithium reserves. Most of the known lithium supply is in Bolivia, Argentina, Chile, Australia and China. It is said that 20 tons of spent Li-ion batteries yield one ton of lithium.What is the name of Li+?
Lithium ion | Li+ - PubChem.Is lithium cation or anion?
As an element, lithium is uncharged, so it is neutral, and neither a cation nor an anion. In aqueous solution, lithium is an Li+ cation, as is typical of Group I elements such as Li, Na, KIs Lithium a compound?
The main lithium compound is the lithium hydroxide. Like all alkali metals, lithium reacts easily in water and does not occur freely in nature due to its activity, Lithium is a moderately abundant element and its present in The Earth's crust in 65 ppm (parts per million).How is a lithium ion formed?
Lithium is in Group 1. It has one electron in its outer shell. When this electron is lost, a lithium ion, Li +, is formed.Is Lithium a metal or nonmetal?
What is lithium, metal, nonmetal, or metalloid? Lithium is a metal, and the lightest metal on the periodic table, with an atomic number of 3. (Just more than hydrogen and helium!)Where is lithium mined?
North America has only one lithium mine, the Albermarle Silver Peak Mine, and only one U.S. company is currently producing lithium from brine. Most of the world's lithium comes from brine operations in Chile and a spodumene operation in Australia. China and Argentina are also major lithium producers.Is lithium reactive?
Reactivity. Lithium is part of the Group 1 Alkali Metals, which are highly reactive and are never found in their pure form in nature. This is due to their electron configuration, in that they have a single valence electron (Figure 1) which is very easily given up in order to create bonds and form compounds.Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?Is Al Cl polar or nonpolar?
Explanation: The Al-Cl bond is polar covalent. This is consistent with the fact that aluminum chloride changes directly from a solid to a gas at the relatively low temperature of 180 °C. Al has three valence electrons.Why is PCl3 polar?
PCl3 is polar, because of its trigonal pyramidal structure. The reflectional asymmetry of the molecule, combined with the fact that chlorine is significantly more electronegative than phosphorus, makes the molecule polar.Is XeF4 polar or nonpolar?
XeF4 Polarity – Is XeF4 Polar or Nonpolar? As we can see that the XeF4 molecular geometry has the symmetric distribution of electrons and they make a formation in the single square plane. So, we can say that the XeF4 is nonpolar.Is AlI3 an acid or base?
Aluminium iodide. Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). is a strong Lewis acid and will absorb water from the atmosphere.What does dipole moment mean?
A dipole moment is a measurement of the separation of two opposite electrical charges. Dipole moments are a vector quantity. The magnitude is equal to the charge multiplied by the distance between the charges and the direction is from negative charge to positive charge: μ = q · r.What type of reaction is iodine and aluminum?
Oxidation of finely dispersed aluminium with iodine can be initiated using drops of water. The reaction is strongly exothermic, and the excess iodine vaporises, forming a deep violet vapour.Is scl6 polar?
Scl6 is non-polar because it has 6 bonding pairs and no lone pairs, giving it an octahedral shape.Is AlI3 ionic or covalent?
In the case of aluminium iodide an ionic bond with much covalent character is present. In the AlI3 bonding, the aluminium gains a +3 charge. The large charge pulls on the electron cloud of the iodines.How do you write chemical formulas?
Part 1 Writing Chemical Formulas of Covalent Compounds- Memorize the prefixes for number of atoms.
- Write the chemical symbol for the first element.
- Add the number of atoms as a subscript.
- Write the chemical symbol for the second element.
- Add the number of atoms present as a subscript.
- Practice with some examples.