.
Just so, is dinitrogen trioxide polar or nonpolar?
Answer = N2O3 ( Dinitrogen trioxide ) is Polar What is polar and non-polar? Polar "In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment.
Similarly, is dinitrogen trioxide a covalent compound? Answer and Explanation: Dinitrogen trioxide (N2O3) is a covalent molecule because it has two nonmetals.
Likewise, what type of compound is dinitrogen trioxide?
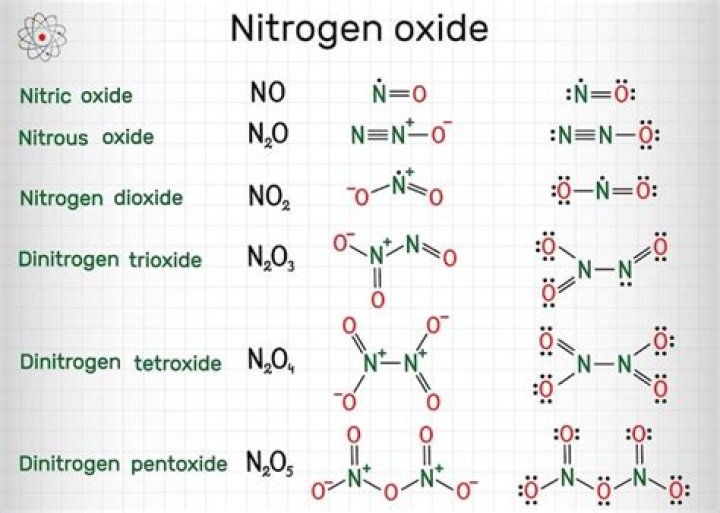
Dinitrogen trioxide is the chemical compound with the formula N2O3. This deep blue solid is one of the simple nitrogen oxides. It forms upon mixing equal parts of nitric oxide and nitrogen dioxide and cooling the mixture below −21 °C (−6 °F): NO + NO2 ⇌ N2O.
What is dinitrogen trioxide used for?
Use as a rocket propellant Nitrogen tetroxide is used as an oxidizer in one of the most important rocket propellants because it can be stored as a liquid at room temperature.
Related Question AnswersIs pcl3 covalent or ionic?
In CCl4, atoms chlorine and carbon share electron pairs to form a covalent bond. Ionic bonds are formed by electrostatic interactions between an electropositive and electronegative atom.Why n2o3 is Coloured?
It is similar to oxygen ( radical pair , paramagnetic with respect to two unpaired electrons) in that sense and liquid oxygen has also blue color but for dimeric O2N-NO2 , the odd electrons on each NO2 get coupled to create the N- N bond and the molecule became diamagentic.Is n2o3 an ionic compound?
Is N2O3 a ionic or covalent compound? An ionic compound comprises of metal and a nonmetal . Therefore NBr3 is a covalent compound , since it is made up of two non-metals .Is nitrogen ionic or covalent?
Nitrogen atoms will form three covalent bonds (also called triple covalent) between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell. Another example of a nonpolar covalent bond is found in the methane (CH4) molecule.Is n2o3 ionic or molecular?
Ionic or Covalent| A | B |
|---|---|
| BaSO4 | Ionic |
| LiF | Ionic |
| NaOH | Ionic |
| N2O3 | Covalent |
What molecular shape does the compound CCl4 have?
tetrahedral moleculeWhat is the formula for nitrogen trioxide?
N2O3What type of compound is dinitrogen monoxide?
Nitrous oxide, also known as dinitrogen oxide or dinitrogen monoxide, is a chemical compound with chemical formula N2O. It is commonly known as laughing gas due to the exhilarating effects of inhaling it.What compound is Si2Br6?
Disilicon Hexabromide Si2Br6 Molecular Weight -- EndMemo.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.Is FeSO4 ionic or covalent?
Identify compound as ionic or covalent then give proper name| Chemical Formula | Type of Compound | Compound Name |
|---|---|---|
| NH3 | covalent | ammonia |
| FeSO4 | ionic | iron (II) sulphate |
| SiO2 | covalent | silicon dioxide |
| GaCl3 | ionic | gallium chloride |