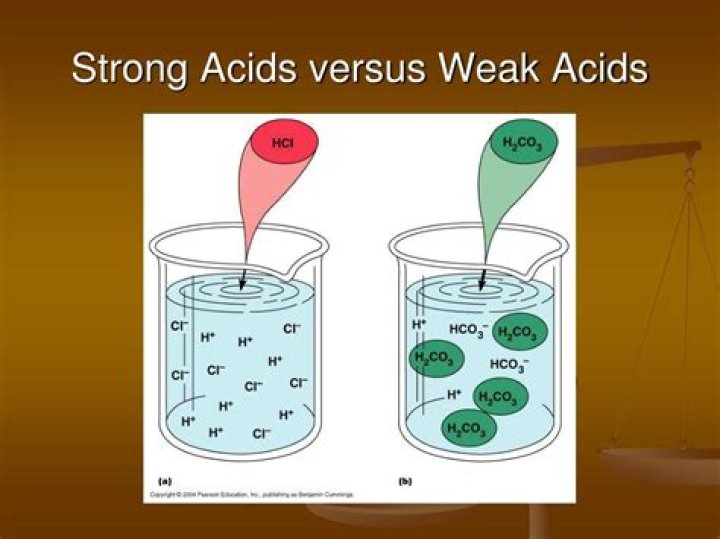
A weak acid is an acid that partially dissociates into its ions in an aqueous solution or water. In contrast, a strong acid fully dissociates into its ions in water. At the same concentration, weak acids have a higher pH value than strong acids..
Similarly one may ask, what determines the strength of an acid?
An acid gets its characteristics from the hydrogen atoms of its molecules. Strong acids have weakly bound hydrogen atoms, and the molecules easily separate from them in solution. How many of these hydrogen atoms dissociate and form hydrogen ions determines the strength of an acid.
One may also ask, what are the 7 strong acids? There are 7 strong acids: chloric acid, hydrobromic acid, hydrochloric acid, hydroiodic acid, nitric acid, perchloric acid, and sulfuric acid. Being part of the list of strong acids doesn't give any indication of how dangerous or damaging an acid is though.
Also, is acetic acid strong or weak?
Acetic acid is a weak acid because it is not a strong acid which has a specific definition in chemistry: Strong acids completely dissociate in aqeous solution, that is, all their H+ come off in water. H+ is also called a proton because hydrogen without an electron is essentially a proton.
Is LiOH strong or weak?
though in the case of lithium, on one hand we have an large electron rich hydroxide ion and on the other is the extremely small, densly packed electron defficient lithium ion with high polarisability leading to a slightly covalent molecule and hence a lower coefficient of dissociation so, LiOH is somewhere in
Related Question Answers
What is strong about a strong acid?
An acid that breaks down completely and gives off many ions, or protons, is considered to be a strong acid. Examples of strong acids include sulfuric acid, hydrochloric acid, perchloric acid, and nitric acid.Is HCl a strong acid?
A strong acid is an acid which is completely ionized in an aqueous solution. Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water. A weak acid is an acid that ionizes only slightly in an aqueous solution. Because HCl is a strong acid, its conjugate base (Cl−) is extremely weak.What are the strong bases?
Strong bases are able to completely dissociate in water - LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.
What are weak acids?
A weak acid is an acid chemical which does not dissociate (split into ions) completely in water solution. This means it does not give all its hydrogen ions into the water. Weak acids typically have a pH between 3 and 6. Acetic acid (CH3COOH) and oxalic acid (H2C2O4) are examples of weak acids.What makes a weak base?
A weak base is a chemical base that does not ionize fully in an aqueous solution. As Brønsted-Lowry bases are proton acceptors, a weak base may also be defined as a chemical base with incomplete protonation.What makes a strong acid or base?
Strong and Weak Acids/Bases[edit] A strong acid is an acid which dissociates completely in water. That is, all the acid molecules break up into ions and solvate (attach) to water molecules. Therefore, the concentration of hydronium ions in a strong acid solution is equal to the concentration of the acid.What is the world's strongest acid?
The carborane superacids may be considered the world's strongest solo acid, as fluoroantimonic acid is actually a mixture of hydrofluoric acid and antimony pentafluoride.Is HCl or HBr stronger?
HBr fumes strongly in moist air. It is one of the strongest mineral acids, with a reducing action stronger than that of hydrogen chloride (HCl). It is extremely soluble in water, forming a strong acid that is available as 48 or 68% solutions.Is nitric acid strong or weak?
All the other acids are weak. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).Is NaOH a strong acid?
Solution: NaOH is a strong base but H2C2O4 is a weak acid since it is not in the table. Therefore, this is a weak acid-strong base reaction which is explained under the link, titration of a weak acid with a strong base.Is HCl stronger than h2so4?
In chemistry, both HCl and H2SO4 are strong acids. But what makes the HCl is stronger than H2SO4 is the difference in Basicity of both acids. In contrast, the HCl is monoprotic acid whereas, the H2SO4 is Diprotic acid. Hence,the number of H+ ions produced by H2SO4 is higher and easily replace with other compounds.Which is the strongest base?
The hydroxide ion is the strongest base possible in aqueous solutions, but bases exist with much greater strengths than can exist in water. Superbases are valuable in organic synthesis and are fundamental to physical organic chemistry. Superbases have been described and used since the 1850s.What is the strength of an acid or base?
The strength of acids and bases depend on how much an acid or base ionizes in solution. A strong acid or base completely ionizes in solution. In a neutralization reaction, an acid and a base react to produce a salt. A salt is an ionic compound whose cation comes from a base and whose anion comes from an acid.Is citric acid a weak or strong acid?
Citric acid solution is a weak acid and is classified as a weak electrolyte. The HCl solution dissociates 100% in water, meaning that all of the HCl reacts with water to form H3O+ ions and Cl- ions. A HCl solution is a strong acid and is classified as a strong electrolyte.Which acid is stronger HCl or h3po4?
Phosphoric acid, when pure, is a solid at room temperature and normal pressure. However, an aqueous solution of 85 % H3PO4 is a viscous liquid. HCl is the strongest acid (indicated by the lowest pKa) followed by H2SO4 and HNO3. H3PO4 is the weakest of the acids.Is HCl a strong electrolyte?
Hydrochloric acid, HCl(aq), dissociates completely so there are many ions in solution to conduct electricity therefore hydrochloric acid is a strong electrolyte. State your solution to the problem "is hydrochloric acid a strong or weak electrolyte": Hydrochloric acid is a strong electrolyte.Is ammonia strong or weak?
Ammonia is a typical weak base. Ammonia itself obviously doesn't contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. However, the reaction is reversible, and at any one time about 99% of the ammonia is still present as ammonia molecules.Is ch3cooh strong?
HCl is a strong acid, and CH3COOH is a weak acid. During a reaction with a strong base, the weak acid is forced to completely dissociate (there is no weak acid molecule left intact at the equivalence point). However, the accepted value for the ∆Hrxn for CH3COOH is about 1.9 kJ/mol less than for the HCl.Is HF stronger than HCl?
HCl is a stronger acid as compared to HF. HCl is a stronger acid as compared to HF. A molecule that produces more protons in a solution is more acidic. If we consider the electronegativity differences, the H-F bond should be weaker and thus cleave easily to give more of H+ in a solution, making HF a stronger acid.