| Element | Symbol | Mass Percent |
|---|---|---|
| Hydrogen | H | 11.190% |
| Oxygen | O | 88.810% |
.
People also ask, what is the percentage of oxygen in h2o?
88.81%
Likewise, what is the percentage composition of h2o? Answer and Explanation: The percent composition of water is 11.19 percent hydrogen and 88.81 percent oxygen.
Herein, how do you calculate the percentage of water?
Divide the mass of the water lost by the mass of hydrate and multiply by 100. The theoretical (actual) percent hydration (percent water) can be calculated from the formula of the hydrate by dividing the mass of water in one mole of the hydrate by the molar mass of the hydrate and multiplying by 100.
How do you calculate the percentage of hydrogen in water?
To find the mass percent of hydrogen in water, take the molar mass of hydrogen in the water molecule, divide by the total molar mass of water, and multiply by 100. Dividing 2.016 by 18.016 gives you 0.1119. Multiply 0.1119 by 100 to get the answer: 11.19 percent.
Related Question AnswersIs oxygen present in water?
Oxygen is the most abundant element on earth. Oxygen exists as O2 and O3 (ozone), and is present in a number of compounds including water molecules. It can be found dissolved in water as O2 molecules. Consequently, the oxygen content of seawater is 85.7%.How much oxygen is in water vs air?
Oxygen concentrations are much higher in air, which is about 21% oxygen, than in water, which is a tiny fraction of 1 percent oxygen. Where the air and water meet, this tremendous difference in concentration causes oxygen molecules in the air to dissolve into the water.How do I calculate moles?
- Start with the number of grams of each element, given in the problem.
- Convert the mass of each element to moles using the molar mass from the periodic table.
- Divide each mole value by the smallest number of moles calculated.
- Round to the nearest whole number. This is the mole ratio of the elements and is.
What percent of 1 mole of water is oxygen?
88.81 percentHow do you calculate weight percent?
Solutions are sometimes represented in terms of relative percent concentration of solute in a solution.To determine the weight percent of a solution, divide the mass of solute by mass of the solution (solute and solvent together) and multiply by 100 to obtain percent.How do you calculate h2o?
Divide the mass of water by the molar mass of water to get moles of water. 2.60 g H2 O / (18.00 g/mol H2 O) = 0.144 moles H2 O. Divide the mass of anhydrate by the molar mass of anhydrate to get moles of anhydrate.How many moles are in h2o?
The average mass of one H2O molecule is 18.02 amu. The number of atoms is an exact number, the number of mole is an exact number; they do not affect the number of significant figures. The average mass of one mole of H2O is 18.02 grams. This is stated: the molar mass of water is 18.02 g/mol.How do you measure oxygen in water?
Measuring dissolved oxygen in water. DO is measured using a dissolved oxygen meter (or DO meter). The best method is to take the measurement in the field at the same time each day, because DO concentrations can vary throughout a 24 hour period.What should my water percentage be?
The average adult human body is 50-65% water, averaging around 57-60%. The percentage of water in infants is much higher, typically around 75-78% water, dropping to 65% by one year of age. Body composition varies according to gender and fitness level because fatty tissue contains less water than lean tissue.What is mass percent of a solution?
Mass percent is used as a way of expressing a concentration or a way of describing a component in a mixture. For a solution, the mass percent is expressed as the grams of solute divided by the grams of solution, then multiplied by 100 to get a percentage.How do you identify an unknown hydrate?
Identify your unknown hydrate from the list of possible hydrated salts below. (atomic mass units) or the mass of a mole of the element in grams/mol. For a compound, the molar mass is defined as the sum of all the masses in one mole of the compound.How do u find the percent error?
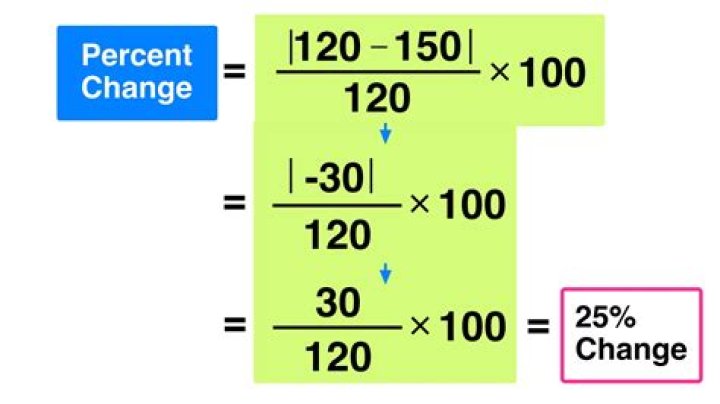
Steps to Calculate the Percent Error- Subtract the accepted value from the experimental value.
- Take the absolute value of step 1.
- Divide that answer by the accepted value.
- Multiply that answer by 100 and add the % symbol to express the answer as a percentage.
What is water weight?
This means that 1 liter (L) of water weighs 1 kilogram (kg) and 1 milliliter (mL) of water weighs 1 gram (g). In common US measures, one gallon of water weighs 8.345 pounds. The density of water varies slightly at different temperatures which will impact the weight of for the same volume.How is total body water measured?
The total body water is then accurately measured from the increase in breath deuterium content in relation to the volume of D2O ingested. Different substances can be used to measure different fluid compartments: total body water: tritiated water or heavy water. extracellular fluid: inulin.Why are hydrates important?
Hydrates are often used in skin care products to infuse moisture into the body. The world contains many gas hydrates, crystalline solids in which gas molecules are enclosed in structures made of water molecules. These form from very low temperatures and high pressure.How much water is in the hydrate?
EXPERIMENTAL MEASUREMENT OF PERCENT HYDRATION: The difference between the two masses is the mass of water lost. Dividing the mass of the water lost by the original mass of hydrate used is equal to the fraction of water in the compound. Multiplying this fraction by 100 gives the percent water in the hydrate.How do I calculate my water weight?
How much water should you drink based on your weight?- Take your weight (in pounds) and divide that by 2.2.
- Multiply that number depending on your age: If you're younger than 30, multiply by 40.
- Divide that sum by 28.3.
- Your total is how many ounces of water you should drink each day.