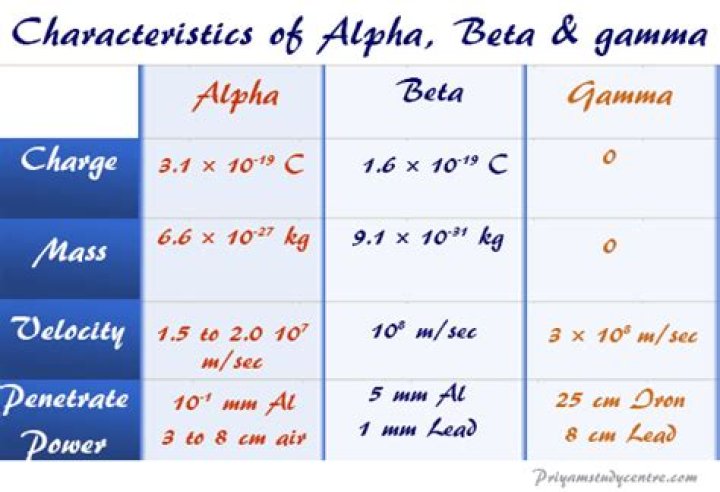
The mass of an alpha particle is 6.645×10−27 kg, while the mass of an electron is 9.11×10−31 kg. Note that you could not solve this by adding together the masses of two protons and two neutrons to get the mass of the alpha, because this would not take into account the binding energy of the particle..
Correspondingly, what is the mass of Alpha Beta and Gamma?
The total mass of an alpha particle is ~4u or 4 unified atomic mass units, equivalent to ~ 6.64 E-27 kg. A beta particle is just an electron, with a mass ritual to around 1/1822 u, or~ 9.1 E-31 kg. A gamma ray is a photon, and therefore massless (no rest mass).
Additionally, what is the mass of an alpha particle in amu? An alpha particle (aka alpha radiation) is basically a Helium nucleus, so 2 ea protons & neutrons, so 4 amu. A beta particle (aka beta radiation) is a high speed electron or positron. Their mass is ~ 1/2000 amu.
Likewise, people ask, how do you find the mass of an alpha particle?
Answer: The alpha particle contains 2 protons and 2 neutrons. Thus it is similar to the Helium atom. Which is equal to 6.68 x 10^-27 kg.
What is the mass of a proton?
Mass of proton: Mass of proton is 1.0072766 a.m.u. or 1.6726 x 10-27 kg. Comparative mass: Proton is 1837 times heavier than an electron. Position in atom: Protons are present in the nucleus of atom.
Related Question Answers
Is Omega higher than Alpha?
Omega Male Definition An omega male is like the opposite of an alpha male, albeit equally cool and confident. Whereas an alpha male is extroverted and the “leader of the pack,” the omega male is more introverted and isn't afraid of doing his own thing and making up his own rules in life.Do gamma rays have mass?
Gamma radiation, unlike alpha or beta, does not consist of any particles, instead consisting of a photon of energy being emitted from an unstable nucleus. Having no mass or charge, gamma radiation can travel much farther through air than alpha or beta, losing (on average) half its energy for every 500 feet.What happens during gamma decay?
Gamma decay, in contrast, occurs when a nucleus is in an excited state and has too much energy to be stable. This often happens after alpha or beta decay has occurred. Because only energy is emitted during gamma decay, the number of protons remains the same.What is the charge of beta?
Beta particles have a charge of minus 1, and a mass of about 1/2000th of a proton. This means that beta particles are the same as an electron.What is Gamma made from?
Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes such as potassium-40, and also as a secondary radiation from various atmospheric interactions with cosmic ray particles.What is an a particle?
Particles are tiny bits of matter that make up everything in the universe. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms and molecules are called microscopic particles. Subatomic particles are particles that are smaller than atoms.How do you make an alpha particle?
An alpha particle is produced by the alpha decay of a radioactive nucleus. Because the nucleus is unstable a piece of it is ejected, allowing the nucleus to reach a more stable state.Where do gamma rays come from?
Gamma rays are often produced in regions of extremely high temperature. Gamma rays come from solar flares (eruptions on the sun's surface), pulsars (rotating objects which regularly emit radiation), nova and super nova explosions (explosions that cause stars to become extremely bright).What are the properties of alpha particles?
Alpha rays are the positively charged particles. Alpha-particle is highly active and energetic helium atom that contains two neutrons and protons. These particles have the minimum penetration power and highest ionization power. They can cause serious damage if get into the body due to their high ionization power.Who discovered Proton?
Rutherford
What are alpha particles Class 9?
Alpha particles are positively charged particles, having two protons and two neutrons and are identical to the nucleus of helium atom. They are spontaneously emitted by radioactive element.What is an alpha particle made of?
An alpha particle is a fast moving packet containing two protons and two neutrons (a helium nucleus). Alpha particles carry a charge of +2 and strongly interact with matter. Produced during alpha decay, alpha particles can travel only a few inches through air and can be easily stopped with a sheet of paper.What is the charge to mass ratio of alpha particle?
Alpha particle is a helium nucleus containing two protons and two neutrons so its charge is t w i c e twice twice the proton's charge while the mass is about 4 times greater. Let the charge of the proton be +e, then the charge of the alpha particle will be +2e.Is a gamma ray a particle?
Gamma rays are electromagnetic radiation, like X-rays. The other two types of natural radioactivity are alpha and beta radiation, which are in the form of particles. Gamma rays are the most energetic form of electromagnetic radiation, with a very short wavelength of less than one-tenth of a nanometer.What happens alpha particles?
Alpha decay occurs when a nucleus is unstable because it has too many protons. The nucleus emits an alpha particle and energy. An alpha particle consists of two protons and two neutrons, which is actually a helium nucleus. Losing the protons and neutrons makes the nucleus more stable.What is alpha beta gamma particles?
The alpha particle is a helium nucleus; it consists of two protons and two neutrons. It contains no electrons to balance the two positively charged protons. Beta particles are negatively charged. Gamma rays are electromagnetic waves of very short wavelength and high frequency.What is the mass of alpha particle and proton?
Since the alpha particle is nothing but positively charged helium nuclei, it has charge of two time of that of the proton, as it is constituted of 2 protons. The rest mass of the alpha particle amounts to 6.64424·10-27 kg, or 3.7273·109 eV, which is almost 4 times of that of proton.What has a mass of 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.What is the mass of deuteron?
But the mass of the deuteron is 1875.6 MeV, implying that, upon energy grounds, it is stable agains such a decay. The free neutron yields an energy of 0.78 MeV in beta decay, but the 2.2 MeV binding energy of the deuteron prevents its decay.