The Hydrophobic Effect. The hydrophobic effect is considered to be the major driving force for the folding of globular proteins. It results in the burial of the hydrophobic residues in the core of the protein. It is exemplified by the fact that oil and water do not mix and was described well by G. S. Hartley in 1936 ..
Also question is, what are hydrophobic interactions in proteins?
Introduction. The hydrophobic (literally, 'water-hating') effect is named for the tendency of certain oil-like substances to avoid contact with water (proverbially, 'oil and water don't mix'). It is generally understood to be the driving force responsible for the folding of proteins.
Secondly, what is the source of the hydrophobic effect? The origin of the hydrophobic effect is not fully understood. Some argue that the hydrophobic interaction is mostly an entropic effect originating from the disruption of highly dynamic hydrogen bonds between molecules of liquid water by the nonpolar solute.
Subsequently, question is, how does the hydrophobic effect work?
The hydrophobic effect describes the energetic preference of nonpolar molecular surfaces to interact with other nonpolar molecular surfaces and thereby to displace water molecules from the interacting surfaces. The hydrophobic effect is due to both enthalpic and entropic effects.
Why is the hydrophobic effect important?
Biological Importance of Hydrophobic Interactions Hydrophobic Interactions are important for the folding of proteins. This is important in keeping a protein stable and biologically active, because it allow to the protein to decrease in surface are and reduce the undesirable interactions with water.
Related Question Answers
Is water hydrophilic?
Nonpolar molecules that repel the water molecules are said to be hydrophobic; molecules forming ionic or a hydrogen bond with the water molecule are said to be hydrophilic. This property of water was important for the evolution of life.What types of proteins are hydrophobic and hydrophilic?
Proteins, made up of amino acids, are used for many different purposes in the cell. The cell is an aqueous (water-filled) environment. Some amino acids have polar (hydrophilic) side chains while others have non-polar (hydrophobic) side chains.Which substance is hydrophobic?
Examples of hydrophobic molecules include the alkanes, oils, fats, and greasy substances in general. Hydrophobic materials are used for oil removal from water, the management of oil spills, and chemical separation processes to remove non-polar substances from polar compounds.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.What is the difference between van der Waals and hydrophobic interactions?
1 Answer. The hydrophobic force arises from the disruption of hydrogen bonds between water molecules, whilst van der Waals interactions are the result of instantaneous and induced dipoles .Is ethanol hydrophobic or hydrophilic?
Ethanol is an interesting molecule. It is polar or hydrophilic (water-loving) due to the presence of the terminal hydroxyl group, so it dissolves in water.What are hydrophilic interactions?
Hydrophilic interactions. When the substrate binds to water then that interaction is known as hydrophilic interaction and the contact angle between water and substrate will be very less. Molecules that have charged parts to them are attracted to the charges within the water molecule.How do hydrophobic interactions affect protein structure?
The Hydrophobic Effect. The hydrophobic effect is considered to be the major driving force for the folding of globular proteins. It results in the burial of the hydrophobic residues in the core of the protein. It is exemplified by the fact that oil and water do not mix and was described well by G. S. Hartley in 1936 .What makes a surface hydrophobic?
A hydrophobic surface is a water repelling, low surface energy surface that resists wetting. Moisture contact angle measurements will classify a surface as hydrophobic when the contact angle of the water droplet exceeds 90 degrees.Why is hydrophilic important?
Since water has these partial charges, it can attract other chemicals that also have partial charges. Therefore, hydrophilic molecules must have a charged portion in order to dissolve in water. Hydrophilicity is an important quality of many essential materials in nature and in the human body.Why does salt increase hydrophobic interactions?
When the salt concentration is increased, some of the water molecules are attracted by the salt ions, which decreases the number of water molecules available to interact with the charged part of the protein. There are hydrophobic amino acids and hydrophilic amino acids in protein molecules.What part of the phospholipid is hydrophobic?
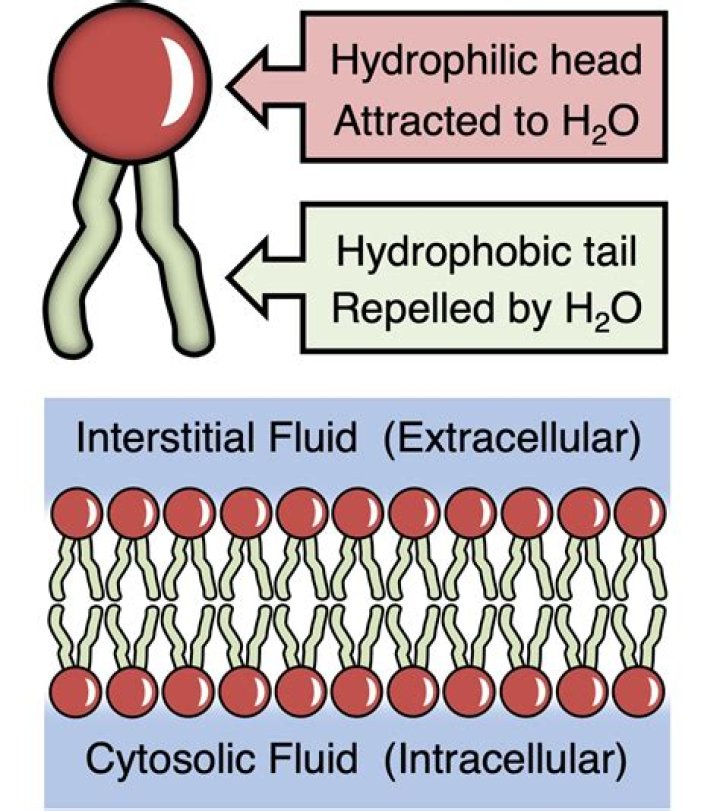
The phosphate group is the negatively-charged polar head, which is hydrophilic. The fatty acid chains are the uncharged, nonpolar tails, which are hydrophobic.Why do hydrophobic molecules repel water?
Hydrophobic literally means “the fear of water”. Hydrophobic molecules and surfaces repel water. The water molecules then form more hydrogen bonds with themselves and the nonpolar molecules clump together. The hydrophobic effect is caused by nonpolar molecules clumping together.Is sodium hydrophilic or hydrophobic?
Examples. Sugar, sodium chloride, and hydrophilic proteins are all substances that dissolve in water. Oils, fats, and certain organic solvents do not dissolve in water because they are hydrophobic.Are enzymes hydrophobic or hydrophilic?
The pink areas are the hydrophilic surface of the enzyme and the blue regions are areas that are hydrophobic. You can see that the active site receives the substrate in an area that is polar to the outside and non-polar to the inside.What is hydrophobic hydration?
Hydrophobic hydration is the hydration of hydrophobic molecules and surfaces.What is hydrophobic in science?
To be hydrophobic means to fear water. In chemistry, it refers to the property of a substance to repel water. Hydrophobic molecules tend to be nonpolar molecules that group together to form micelles rather than be exposed to water. Hydrophobic molecules typically dissolve in nonpolar solvents (e.g., organic solvents).Are esters hydrophobic?
Consequently, esters and ketones bearing typical polar groups are not classified into hydrophilic compounds, but into “hydroneutral” compounds positioned between hydrophilic and hydrophobic ones.Why is hydrophobicity important to an amino acid?
When a protein folds, hydrophobic amino acids get buried inside the protein to form a hydrophobic core. The tendency of hydrophobic groups to cluster together when they are put into water—or the hydrophobic effect—is the most important driving force in protein folding.