.
Similarly one may ask, what is the hybridization of the chlorine atom in ClF5?
1 Answer. Stefan V. The hybridization of the chlorine atom in ClF5 is sp3d2 .
Likewise, what is the hybridization of the central atom in xecl2? The arrangement of the electrons of Xenon changes to s2 p5 d1 with two unpaired electrons. Hence the hybridization of the central atom Xe is sp3d.
Consequently, what is the Lewis structure for ClF5?
For ClF5 Cl goes in the center since it is the least electronegative. You will need to place the remaining two valence electrons on the central Cl after filling the octets of the Fluorine atoms. For the ClF5 Lewis structure there are a total of 42 valence electrons available.
Is ClF5 polar or non polar?
It appears to be asymmetrical, and is polar. The geometry is square pyramidal and is due to 6 electrons pairs around the central chlorine atom, one of which is nonbonding. A Cl-F bond is a dipole since F is more electronegative.
Related Question AnswersHow do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
Why is ClF polar?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). For these molecules, the applicable intermolecular interactions are dipole and induced dipole interactions.What is the shape of ClF5?
The molecular geometry of ClF5 is square pyramidal with asymmetric charge distribution around the central atom.What is the bond order of cl2?
the shortest bond because it has a bond order of 1. 5.3 a. diatomics from the row directly above them in the periodic table: P2 bond order = 3 (like N2) S2 bond order = 2 (like O2) Cl2 bond order = 1 (like F2) Cl2 has the weakest bond.What is the hybridization of central carbon in 1/2 propadiene?
Dear Student, When a carbon is attached with two carbons by two double bonds is called allene. Allene is 1,2 propadiene. The central carbon is sp hybridised while the terminal carbons are sp2 hybridised.Does ClF5 have a dipole moment?
Both of these have an sp3d2 hybrid about the central atom. ClF5 , however, is out of balance, and therefore does have a dipole.How many σ and π bonds are present in a molecule of Cumulene?
Cumulene structure shows sigma bond on the end-side carbon with hydrogen. There are four total sigma bond of carbon to hydrogen. 3 Sigma bonds and 3 pi bonds connect carbon to carbon in middle. That makes total 7 sigma bonds and 3 pi bonds in cumulene.What is the hybridization of the central atom of pbr5?
As one electron goes to s orbital, three occupy the p orbital, and the last one enters the d orbitals of the central atom, the hybridization of Pbr5 is sp3d.How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.Is SeO2 polar or nonpolar?
The three bond dipoles from the three polar Se-O bonds in SeO3 will all cancel when summed together. Hence, SeO3 is nonpolar since the overall molecule has no resulting dipole moment. In SeO2, the two Se-O bond dipoles do not cancel when summed together, hence SeO2 has a dipole moment ( is polar).How is XeF2 formed?
Xenon difluoride(XeF2) It is prepared by heating a mixture of Xenon and fluorine in the ration 2:1 at 400 degree Celsius and 1 bar pressure in a sealed nickel tube. XeF2 undergoes hydrolysis when treated with water an d evolves oxygen. In XeF2, Xenon is sp3d hybridised and the molecule has linear structure as shown.Is CCl4 polar or nonpolar?
CCl4 is an example of a nonpolar molecule. The four bonds of carbon tetrachloride (CCl4) are polar, but the molecule isnonpolar because the bond polarity is canceled by the symmetric tetrahedral shape. When other atoms substitute for some of the Cl atoms, the symmetry is broken and the molecule becomes polar.Is no2 linear?
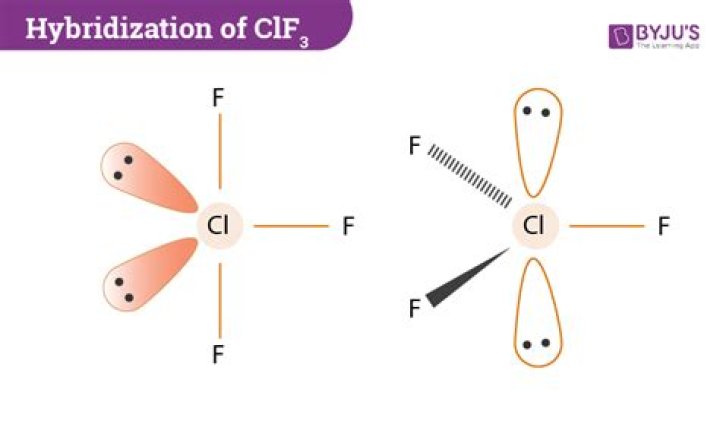
Just like carbon dioxide? Except that NO2 is a V-shaped molecule, and CO2 is linear. two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.Is clf3 polar?
The molecular geometry of ClF3 is T-shaped with asymmetric charge distribution around the central atom. Therefore ClF3 is polar.Why is PCl5 nonpolar?
for example Cl is more electronegative than P, thus the bonds in PCl5 are polar, but the molecule overall is nonpolar due to the arrangement of Cl atoms in a trigonal bipyramidal structure where they share electrons equally.How do I calculate bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order:- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.