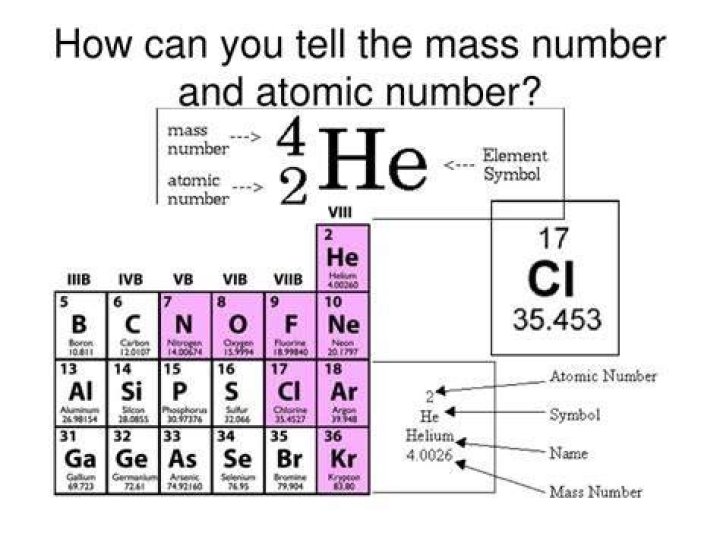
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus..
Keeping this in consideration, what is atomic number and atomic mass?
A property closely related to an atom's mass number is its atomic mass. The atomic mass of a single atom is simply its total mass and is typically expressed in atomic mass units or amu. By definition, an atom of carbon with six neutrons, carbon-12, has an atomic mass of 12 amu.
Also Know, what does the atomic mass represent? Each element has a specific mass number and a specific atomic number. These two numbers are fixed for an element. The mass number tells us the number (the sum of nucleons) of protons and neutrons in the nucleus of an atom.
Consequently, what is atomic number in chemistry?
Atomic number is the number of protons, and therefore also the total positive charge, in the atomic nucleus. The Rutherford–Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1).
Is atomic mass and mass number the same?
Atomic mass is the weighted average mass of an atom of an element based on the relative natural abundance of that element's isotopes. The mass number is a count of the total number of protons and neutrons in an atom's nucleus.
Related Question Answers
How is atomic mass calculated?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. You can see from the periodic table that carbon has an atomic number of 6, which is its number of protons. The atomic mass of the atom is the mass of the protons plus the mass of the neutrons, 6 + 7, or 13.Who discovered atomic mass?
John Dalton
What is atomic mass in science?
Atomic mass or weight is the average mass of the protons, neutrons, and electrons in an element's atoms.What has a mass of 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.Is the mass number on the periodic table?
Mass Number. On the periodic table, the mass number is usually located below the element symbol. The mass number listed is the average mass of all of the element's isotopes.What makes up the atomic mass?
Atomic mass is defined as the number of protons and neutrons in an atom, where each proton and neutron has a mass of approximately 1 amu (1.0073 and 1.0087, respectively). The electrons within an atom are so miniscule compared to protons and neutrons that their mass is negligible.Why is the atomic mass a decimal?
Though individual atoms always have an integer number of atomic mass units, the atomic mass on the periodic table is stated as a decimal number because it is an average of the various isotopes of an element.What is atomic number Simple?
noun. the number of positive charges or protons in the nucleus of an atom of a given element, and therefore also the number of electrons normally surrounding the nucleus. Abbreviation: at. no.; Symbol: Z.What is number 4 on the periodic table?
The element with atomic number 4 is beryllium, which means each atom of beryllium has 4 protons. A stable atom would have 4 neutrons and 4 electrons. Varying the number of neutrons changes the isotope of beryllium, while varying the number of electrons can make beryllium ions.Whats is an atom?
An atom a fundamental piece of matter. An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.What is the other name of atomic number?
Also Known As: The atomic number is also known as the proton number.WHAT IS A in chemistry?
A is used to signify the atomic mass number (also known as atomic mass or atomic weight) of an atom. A = # protons + # neutrons. A and Z are integer values. When the actual mass of an atom is expressed in amu (atomic mass units) or g/mol then the value is close to A.What is atomic number example?
Examples of Atomic Numbers If an atom has one proton then the atomic number is 1 and the element is hydrogen. Every carbon atom will have 6 protons and the atomic number of carbon is 6. Every cesium atom has 55 protons and hence the atomic number is 55.What is the atomic weight?
Atomic weight is the average mass of atoms of an element, calculated using the relative abundance of isotopes in a naturally-occurring element. It is the weighted average of the masses of naturally-occurring isotopes.What do you mean by Valency?
In chemistry, the valence or valency of an element is a measure of its combining power with other atoms when it forms chemical compounds or molecules. The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds.What is atomic mass give example?
Examples of Atomic Mass Remember, electrons are too small to be considered in the atomic mass. Helium is the second lightest element, and is larger than hydrogen. It consists of two protons and two neutrons. Because protons and neutrons are approximately the same size, the atomic mass of a helium atom is four daltons.Why is Atomic Mass important?
Atomic mass is extremely important in chemistry because it is the connection between mass, which we can measure in the laboratory, and moles, which are numbers of atoms. Most of what we study in chemistry is determined by ratios of atoms.Why is the atomic mass in a fraction?
The atomic masses of most elements are fractional because they exist as a mixture of isotopes of different masses. Most elements occur as a mixture of isotopes of different masses. The fractional atomic masses arise because of this mixture. mass = total mass of all atoms/number of atoms.How was atomic mass first measured?
Rather, these first measurements were made by comparing weights of various atoms to hydrogen. Hydrogen was chosen as the unit of comparison because it was the lightest substance known and the weights of the other elements would be very close to whole numbers.