.
Also to know is, what is a single covalent bond Why does it form?
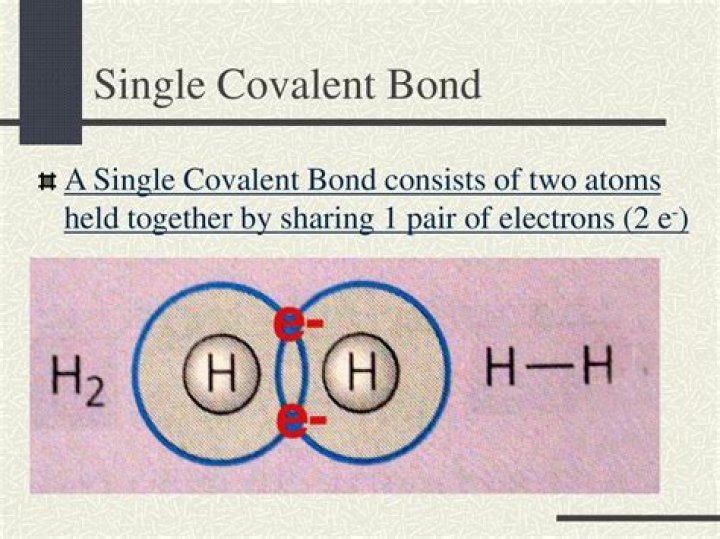
Single Covalent Bonds. A covalent bond form when two orbitals with one electron each overlap each other. Each F atom also has three pair of electrons that are not shared with the other atom. A lone pair is a pair of electrons in a Lewis electron-dot structure that is not shared between atoms.
Furthermore, what is meant by covalent bond? A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
Herein, how is a single covalent bond formed?
When a covalent bond forms with one pair of shared electrons, we call it a single covalent bond. When two pairs are shared, as when two oxygen atoms come together to make a molecule of oxygen, we call it a double covalent bond. This means that each carbon atom can form four single covalent bonds with four other atoms.
What is a nonpolar covalent bond quizlet?
The energy required to break the bonds in one mole of a chemical compound. nonpolar covalent bond. A covalent bond in the bonding electrons are equally attracted to both atoms. polar covalent bond. A covalent bond in which a shared pair of electrons is held more closely by one of the atoms.
Related Question AnswersWhat is another name for a single covalent bond?
Another name for single covalent bonds. Sigma bonds. Sigma bonds. A single covalent bond that if formed when an electron pair is shared by the direct overlap of bonding orbitals and the area is centered between the two atoms. When two atoms share electrons what's overlaps.Is o2 a single covalent bond?
Two chlorine atoms can share 1 electron each to form a single covalent bond. Oxygen can also form covalent bonds, however, it needs a further 2 electrons to complete its valence shell (it has 6). Two oxygen atoms must share 2 electrons each to complete each other's shells, making a total of 4 shared electrons.Is water a single covalent bond?
Water is not a covalent bond. Rather, the bonds in H2O are covalent. And so, H2O is a simple covalent molecule. The oxygen atom shares an outermost electron each with one hydrogen atom (which also shares an outermost electron) to give a single bond.What are the three types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.What is the difference between a sigma bond and a pi bond?
A sigma bond is your basic head-on covalent bond, with the bond in line with the bonding orbitals. You can only ever have one sigma bond between any two atoms. A pi bond is a covalent bond between orbitals perpendicular to the bond direction, usually p-orbitals (never s). A triple bond is one sigma and two pi bonds.Which type of bond is the strongest?
The strongest chemical bond is the covalent bond.- but theres no specific answer to this question because there exist various facters on which it depends like bond enthalpy.
- Stability of the atoms after break up of the bonds and many more.
How many electrons are in a triple bond?
One covalent bond contains 2 electrons, one from each atom. A triple bond indicates that, well, there are three total covalent bonds in that one bond. So, 3 bonds x 2 electrons per bond would give you 6 total electrons.How coordinate bond is formed?
A coordinate bond is formed when a ligand molecule (or ion) that has extra lone pairs on it uses those pairs to join another atom that has empty valence orbitals. The bond that is formed is the lone pair sharing into the empty orbital of the second atom.Does co2 have a single covalent bond?
Answer and Explanation: There are four covalent bonds in one molecule of carbon dioxide. Carbon dioxide consists of one carbon molecule and two oxygen molecules.How a covalent bond is formed?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.What determines the strength of a covalent bond?
The strength of a covalent bond is measured by its bond dissociation energy, that is, the amount of energy required to break that particular bond in a mole of molecules. Multiple bonds are stronger than single bonds between the same atoms.How do you identify a covalent bond?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.Do all the molecules contain only single bonds?
A single bond is defined as the bond through which two same or different atoms are combined to each other by a single line. It is represented by a hyphen. It is not necessary that all the molecules contain a single bond. This is because there are molecules which contain a double or triple bond also.What is a single covalent bond quizlet?
Single Covalent Bond. a bond formed when two atoms share a single pair of electrons. Structural Formula. a chemical formula that shows the arrangement of atoms in a molecule or a polyatomic ion; each dash between a pair of atoms indicated a pair of shared electrons. Unshared Pair.What is the difference between a single and double covalent bond?
Single covalent bond involves both atoms sharing one atom which means there are two electrons in the bond. However, in a double covalent bond each atom shares two electrons which means there are 4 electrons in the bond.How many electrons are shared in a single covalent bond?
two electronsWhat are some examples of covalent bonds?
Examples of compounds that contain only covalent bonds are methane (CH4), carbon monoxide (CO), and iodine monobromide (IBr). Covalent bonding between hydrogen atoms: Since each hydrogen atom has one electron, they are able to fill their outermost shells by sharing a pair of electrons through a covalent bond.Is h2o a covalent bond?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.What are the characteristics of covalent bonds?
Properties of Covalent Molecular Compounds.- Low melting points and boiling points.
- Low enthalpies of fusion and vaporization These properties are usually one or two orders of magnitude smaller than they are for ionic compounds.
- Soft or brittle solid forms.
- Poor electrical and thermal conductivity.