.
Also to know is, what is PEM in chemistry?
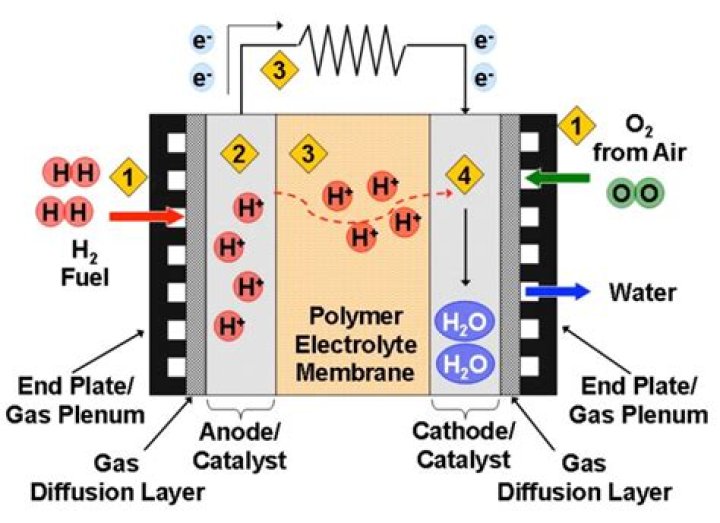
Proton-exchange membrane fuel cells, also known as polymer electrolyte membrane (PEM) fuel cells (PEMFC), are a type of fuel cell being developed mainly for transport applications, as well as for stationary fuel-cell applications and portable fuel-cell applications.
One may also ask, what is the role of membrane in PEM fuel cell? Membranes. In order for a PEM fuel cell to operate, a Proton Exchange Membrane is needed that will carry the hydrogen ions, proton, from the anode to the cathode without passing the electrons that were removed from the hydrogen atoms.
Simply so, how does a proton exchange membrane work?
The proton exchange membrane fuel cell (PEMFC) uses a water-based, acidic polymer membrane as its electrolyte, with platinum-based electrodes. The protons pass through the membrane to the cathode side of the cell while the electrons travel in an external circuit, generating the electrical output of the cell.
What are the four main parts of a PEM?
The membrane, catalyst layers (anode and cathode), and diffusion media together form the membrane electrode assembly (MEA) of a PEM fuel cell.
Related Question AnswersWhat are good things about hydrogen?
Hydrogen has three basic benefits that address these concerns. The use of hydrogen greatly reduces pollution. When hydrogen is combined with oxygen in a fuel cell, energy in the form of electricity is produced. This electricity can be used to power vehicles, as a heat source and for many other uses.Why is Nafion used in fuel cells?
Nafion Membranes are widely used in fuel cells because of their high ionic conductivity. Nafion is highly resistant to chemical attack (corrosion resistant) making it a stable option for use in fuel cells. When used as an ion exchange membrane it is specified to operate at temperatures up to 190°C.How do PEM electrolyzers work?
In a polymer electrolyte membrane (PEM) electrolyzer, the electrolyte is a solid specialty plastic material. Water reacts at the anode to form oxygen and positively charged hydrogen ions (protons). The electrons flow through an external circuit and the hydrogen ions selectively move across the PEM to the cathode.How do fuel cells work?
A fuel cell works by passing hydrogen through the anode of a fuel cell and oxygen through the cathode. At the cathode, the protons, electrons, and oxygen combine to produce water molecules. Due to their high efficiency, fuel cells are very clean, with their only by-products being electricity, excess heat, and water.How is hydrogen made?
As of 2018, the majority of hydrogen (∼95%) is produced from fossil fuels by steam reforming of natural gas, partial oxidation of methane and coal gasification. Other methods of hydrogen production include biomass gasification and electrolysis of water.What are the different types of fuel cells?
Types of Fuel Cells- Polymer electrolyte membrane fuel cells.
- Direct methanol fuel cells.
- Alkaline fuel cells.
- Phosphoric acid fuel cells.
- Molten carbonate fuel cells.
- Solid oxide fuel cells.
- Reversible fuel cells.
What is a fuel cell stack?
A single fuel cell consists of a membrane electrode assembly (MEA) and two flow-field plates delivering about 0.5 and 1V voltage (too low for most applications). Just like batteries, individual cells are stacked to achieve a higher voltage and power. This assembly of cells is called a fuel cell stack, or just a stack.What are alkaline fuel cells used for?
Alkaline fuel cells (AFCs) are one of the most developed technologies and have been used since the mid-1960s by NASA in the Apollo and space shuttle programs. The fuel cells on board these spacecraft provide electrical power for onboard systems, as well as drinking water.What are the disadvantages of fuel cells?
Disadvantages:- Expensive to manufacture due the high cost of catalysts (platinum)
- Lack of infrastructure to support the distribution of hydrogen.
- A lot of the currently available fuel cell technology is in the prototype stage and not yet validated.
- Hydrogen is expensive to produce and not widely available.