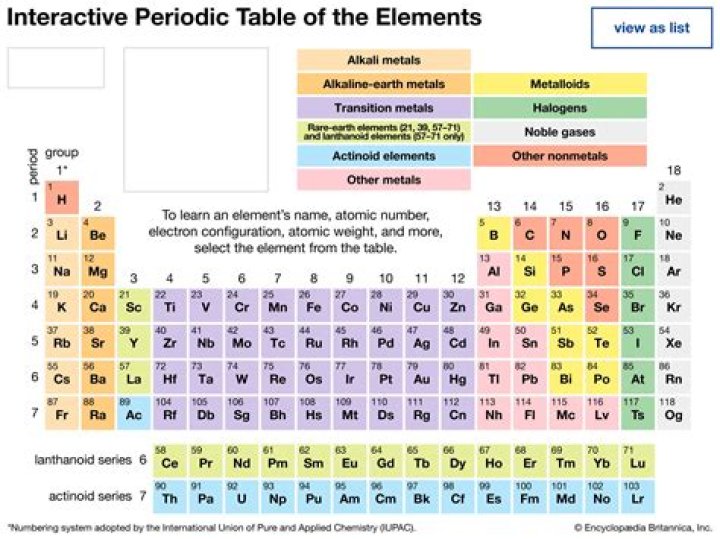
The periodic table also has a special name for its vertical columns. Each column is called a group. The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons..
Herein, what are Group A elements known as?
Elements with similar reactivity are put into the same column or group. The elements in group IA are called the alkali metals. The elements in group IIA are called the alkaline earth metals. The elements in group VIIA are called the halogens and the elements in group VIIIA are called the noble gases or the inert gases.
Also Know, what are Group 3 elements called? The four group 3 elements are scandium, yttrium, lanthanum and actinium. “lanthanide” refers to the elements between lanthanum and lutetium. That term does not encompass scandium and yttrium. Scandium and yttrium act pretty much identically to lanthanides.
Similarly, you may ask, what are Group B elements called?
Group B metals are referred to as transition metals. They are located in the middle of the periodic table between Group IIA and Group IIIA.
What is another name for groups on the periodic table?
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table; the f-block columns (between groups 3 and 4) are not numbered.
Related Question Answers
Which group is least reactive?
Group 18: Noble Gases Group 18 elements are nonmetals called noble gases (see Figure below). They are all colorless, odorless gases. Their outer energy level is also full, so they are the least reactive elements.Who discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.How many types of elements are there?
There are more than 109 different types of atom - one for each element. Differences between the atoms give the elements their different chemical properties. In 2001, there were 115 known elements. However, those above 109 are highly unstable and have been made in only tiny quantities.What are Group 17 elements called?
The halogens are located on the left of the noble gases on the periodic table. These five toxic, non-metallic elements make up Group 17 of the periodic table and consist of: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At).What is Group 13 on the periodic table called?
Group 13 is sometimes referred to as the boron group, named for the first element in the family. These elements are--not surprisingly--located in column 13 of the periodic table. This group includes boron, aluminum, gallium, indium, thallium, and ununtrium (B, Al, Ga, In, Tl, and Uut, respectively).What are the main elements?
The Five Basic Elements are Fire, Earth, Water, Metal, and Wood.What group is the most reactive nonmetals?
Answer and Explanation: The most reactive nonmetal is fluorine. Fluorine is a halogen, which is Group 17 on the periodic table, and the halogens are the most reactiveWhat is Group A in the periodic table?
A group is any column on the periodic table. Elements in the same group usually have similar properties, because they have the same number of electrons in the outermost electron shell. There are eight main groups of elements, numbered 1, 2, and 13-18.Are halogens metals?
The halogens are a series of non-metal elements from group 17 of the periodic table (formerly VII). The halogens include fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The artificially created element 117 (ununseptium) may also be considered a halogen.Which is the most reactive metal?
cesium
Why are Group B called transition metals?
The transition metals were given their name because they had a place between Group 2A (now Group 2) and Group 3A (now Group 13) in the main group elements. Therefore, in order to get from calcium to gallium in the Periodic Table, you had to transition your way through the first row of the d block (Sc → Zn).Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.Are halogens reactive?
The halogens are all elements that are found in group 17 of the periodic table. The halogens include fluorine, chlorine, bromine, iodine, and astatine. All of these elements are considered to be reactive nonmetals. Because these atoms are so close to having a full set of eight valence electrons, they're very reactive.Why are they called alkaline earth metals?
They are called alkaline earth metals because they form alkaline solutions (hydroxides) when they react with water . So basically, this term alkaline means that the solution has a pH greater than seven and is basic. Periodic Table: Why are group 2A elements called alkaline earth metals?How many metals are there?
About 91 of the 118 elements in the periodic table are metals; the others are nonmetals or metalloids. Some elements appear in both metallic and non-metallic forms.Is Lithium a metal?
Lithium. Lithium is part of the alkali metal group and can be found in the first column of the periodic table right below hydrogen. Like all alkali metals it has a single valence electron that it readily gives up to form a cation or compound. At room temperature lithium is a soft metal that is silvery-white in color.Are alkali metals or halogens more reactive?
Reactivity of halogens and alkali metals. As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive. As you go up group 7 (the halogens), again, the elements become more reactive.Is group 3 metal or nonmetal?
The elements in group 3 of the periodic table are boron, aluminum, gallium, indium and thallium. Elements can be classified as metals or non-metals.How many elements are there in 3rd group?
four