.
In respect to this, which group in the periodic table is known as the halogens?
Group 17
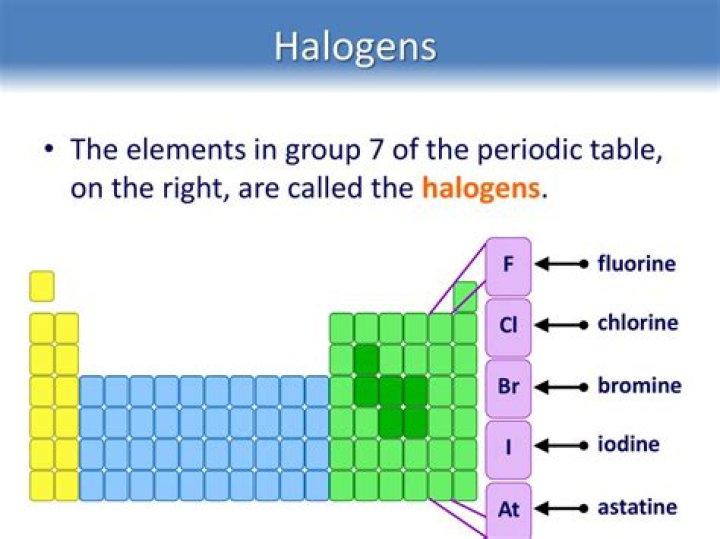
Also Know, what is Group 7 on the periodic table called? The Group 7 elements are called the halogens. They are placed in the vertical column, second from the right, in the periodic table . Chlorine, bromine and iodine are the three common Group 7 elements. Group 7 elements form salts when they react with metals.
Regarding this, why are Group 7 called halogens?
Group 7 – The Halogens. The elements in Group 7 of the Periodic Table are all non-metals. They have been given the name halogens from the Greek words meaning 'salt makers'. The salt in your kitchen, sodium chloride, is a compound of the halogen chlorine.
What is halogen group in chemistry?
The halogens are five non-metallic elements found in group 17 of the periodic table. The term "halogen" means "salt-former" and compounds containing halogens are called "salts". All halogens have 7 electrons in their outer shells, giving them an oxidation number of -1.
Related Question AnswersWho discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.Which element is most reactive?
Fluorine is identified as the most electronegative element in the periodic table, making it the strongest oxidizing agent. It is the most reactive non-metal. Fluorine is so reactive that it can burn substances that one would generally think of as non-flammable!Why halogens are called halogens?
Group 17 elements are called halogens because halogen is a Greek word which means 'salt producing'. They react with metals to form compounds called salts.Are halogens flammable?
Hydrogen halides are formed during combustion of halogen-containing organic compounds. colourless, non-flammable, toxic gas with a pungent smell. Very soluble in water, forming hydrochloric acid and thus a highly corrosive fog in wet air.What is Group 0 called?
Physical properties of the noble gases Group 0 contains non-metal elements placed in the vertical column on the far right of the periodic table . The elements in group 0 are called the noble gases . They exist as single atoms .Are halogens metals?
The halogens are a series of non-metal elements from group 17 of the periodic table (formerly VII). The halogens include fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The artificially created element 117 (ununseptium) may also be considered a halogen.What are halogens elements?
Halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).What is Group 18 called?
Noble Gases[edit] The noble gases are in Group 18 (8A). They are helium, neon, argon, krypton, xenon, and radon. They were once called inert gases because they were thought to be completely inert—unable to form compounds.What are halogens called?
The elements of Group VIIA (new Group 17 – fluorine, chlorine, bromine, iodine, and astatine) are called the halogens (tan column). The term “halogen” means “salt-former” because these elements will readily react with alkali metal and alkaline earth metals to form halide salts.Are halogens reactive?
The halogens are all elements that are found in group 17 of the periodic table. The halogens include fluorine, chlorine, bromine, iodine, and astatine. All of these elements are considered to be reactive nonmetals. Because these atoms are so close to having a full set of eight valence electrons, they're very reactive.Which group is called Chalcogens?
The chalcogens (/ˈkælk?d??nz/) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. It consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive element polonium (Po).What is Group 8 called?
Group 8 is a group (column) of chemical elements in the periodic table. It consists of iron (Fe), ruthenium (Ru), osmium (Os) and hassium (Hs). They are all transition metals.Why are the halogens diatomic?
Halogens are diatomic because they have an outer shell of seven valence electrons and react with other atoms of the same element to complete theirIs Xe a halogen?
The halogens or halogen elements; fluorine (F), chlorine (Cl), bromine (Br), Iodine (I), astatine (At) and possibly the unknown (Uss), are a group of nonmetal elements.Halogens.
| 50 Sn 118.7 |
| 51 Sb 121.8 |
| 52 Te 127.6 |
| 53 I 126.9 |
| 54 Xe 131.3 |