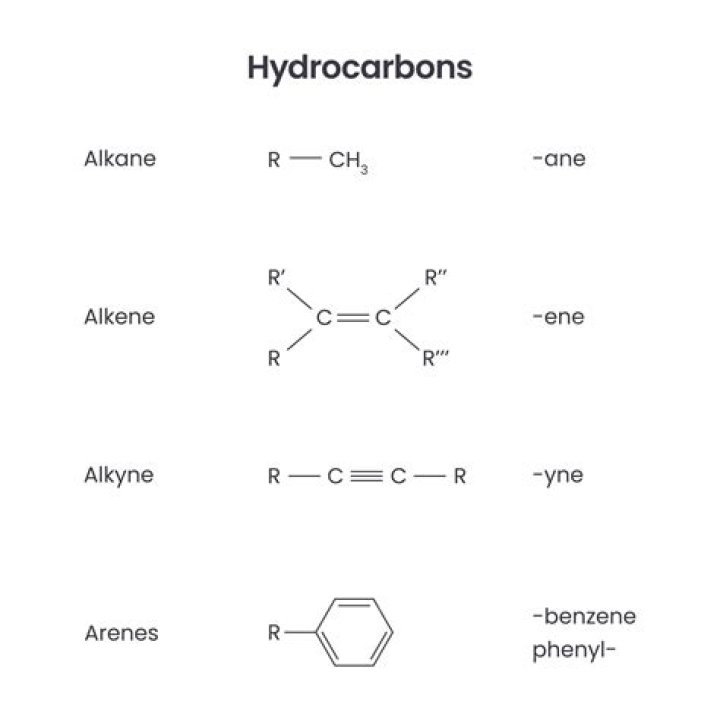
A hydrocarbon is any of a class of organic chemicals made up of only the elements carbon (C) and hydrogen (H). The carbon atoms join together to form the framework of the compound, and the hydrogen atoms attach to them in many different configurations.
What are characteristics of hydrocarbons?
Hydrocarbons are organic compounds composed of only carbon and hydrogen. The alkanes are saturated hydrocarbons—that is, hydrocarbons that contain only single bonds. Alkenes contain one or more carbon-carbon double bonds. Alkynes contain one or more carbon-carbon triple bonds.
Which of the following best describes hydrocarbons?
A hydrocarbon is an organic compound consisting of hydrogen and carbon found in crude oil, natural gas, and coal. Hydrocarbons are highly combustible and the main energy source of the world. Its uses consist of gasoline, jet fuel, propane, kerosene, and diesel, to name just a few.
What is typical for hydrocarbons?
A hydrocarbon is an organic compound consisting of only hydrogen and carbon atoms. They are group 14 hybrids, which means they contain hydrogen, as well as atoms of the carbon 14 group; carbon, silicon, germanium, tin, and lead. … Hydrocarbons can form more complex compounds, like cyclohexane, by bonding to themselves.What is special about hydrocarbons?
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic with only weak odours. Because of their diverse molecular structures, it is difficult to generalize further.
What are the key characteristics of a hydrocarbon combustion reaction?
Hydrocarbon combustion refers to the chemical reaction where a hydrocarbon reacts with oxygen to create carbon dioxide, water, and heat. Hydrocarbons are molecules consisting of both hydrogen and carbon.
How do you identify a hydrocarbon?
- The ene suffix (ending) indicates an alkene or cycloalkene.
- The longest chain chosen for the root name must include both carbon atoms of the double bond.
- The root chain must be numbered from the end nearest a double bond carbon atom.
What is the most common classification of hydrocarbons?
Alkanes are hydrocarbons in which all of the bonds are single bonds. Alkenes are hydrocarbons that contain a carbon-carbon double bond. Alkynes are hydrocarbons that contain a carbon-carbon triple bond. The most common aromatic hydrocarbons are those that contain a benzene ring.What does a hydrocarbon contain?
hydrocarbon, any of a class of organic chemical compounds composed only of the elements carbon (C) and hydrogen (H). The carbon atoms join together to form the framework of the compound, and the hydrogen atoms attach to them in many different configurations.
What are the characteristics of organic compounds?- Organic compounds include complex structures and high molecular weights.
- These are soluble in organic solvents and mostly insoluble in water.
- Mostly depend on only three elements: Carbon, Hydrogen and nitrogen.
- These compounds are combustible in nature.
What is hydrocarbon and its classification?
Hydrocarbons are organic compounds that contain only carbon and hydrogen. The four general classes of hydrocarbons are: alkanes, alkenes, alkynes and arenes.
What is the definition of hydrocarbon quizlet?
Hydrocarbon. the simplest organic compounds which are made of only hydrogen and carbon atoms.
What are hydrocarbons explain with example?
(a) Hydrocarbon is a compound of hydrogen and carbon. Examples for hydrocarbons are methane (CH4) , ethane (C2H6)and ethyne (C2H2). All these compounds are hydrocarbons as they are only made of hydrogen and carbon elements which is evident from their respective molecular formulae.
What elements do hydrocarbon fuels sometimes contain?
Fuels such as natural gas and petrol contain hydrocarbons. These are compounds of hydrogen and carbon only.
How are hydrocarbons used in everyday life?
We use hydrocarbons every day, mainly as fuels, such as natural gas, acetylene, propane, butane, and the principal components of gasoline, diesel fuel, and heating oil. … We can distinguish several types of hydrocarbons by differences in the bonding between carbon atoms.
Why are hydrocarbons good fuels?
Hydrocarbons are organic molecules consisting entirely of carbon and hydrogen. … They make good fuels because their covalent bonds store a large amount of energy, which is released when the molecules are burned (i.e., when they react with oxygen to form carbon dioxide and water).
What do hydrocarbons look like?
A hydrocarbon is an organic compound made of nothing more than carbons and hydrogens. … Saturated hydrocarbons have as many hydrogen atoms as possible attached to every carbon. For carbons on the end of a molecular chain, three can be attached. For carbons in the middle of a chain or a ring, two can be attached.
What are the 4 types of hydrocarbons?
Hydrocarbon molecules have one or more central carbon atoms in a branched or chain-like structure, surrounded by hydrogen atoms. There are four main categories of hydrocarbons: Alkanes, Alkenes, Alkynes, and Aromatic hydrocarbons.
Why are hydrocarbons reactive?
Hydrocarbons that have a double bond between two carbon atoms are called alkenes. These are more reactive than alkanes. Electron-poor molecules react at the pi bond of alkenes. … Electron-poor molecules react at the pi bond of alkynes.
Why are hydrocarbons flammable?
Hydrocarbons are flammable because the carbon in their structure is in the most reduced form.
How are hydrocarbons prepared?
Preparation of Hydrocarbons – Alkanes The alkanes can be produced from alkenes or alkynes through hydrogenation. H2 gas is passed over a metal surface such as Ni, Pt along with the alkenes to produce alkane. The above reaction is called “Sabatier-Sender son’s” reaction.
What hydrocarbons are gases and liquids?
- Alkanes, or paraffins. Ethane—C2H6 Propane—C3H8 Butanes: normal butane and isobutane—C4H10 Natural gasoline or pentanes plus—C5H12 and heavier.
- Alkenes, or olefins. Ethylene—C2H44. Propylene—C3H6 Normal butylene and isobutylene—C4H8
What are characteristics of inorganic compound?
An inorganic compound can be considered as a compound that does not contain a carbon-to-hydrogen bond, also called a C-H bond. Moreover, inorganic compounds tend to be minerals or geologically-based compounds that do not contain carbon-to-hydrogen bonds. Not all, but most inorganic compounds contain a metal.
What are the four characteristic of organic compound?
- 1 . They all contain carbon.
- 2 . Most of them are flammable.
- 3 . They are all soluble in non-polar solvents.
- 4 . They are most, if not all, are covalently bonded molecules.
What are 4 characteristics of organic molecules?
These atoms’ ability to attach to one another allows for the creation of innumerable compounds conducive to life. All organisms need four types of organic molecules: nucleic acids, proteins, carbohydrates and lipids; life cannot exist if any of these molecules are missing.
What are hydrocarbons class 9th?
Hydrocarbons are organic compounds composed of elements of carbon and hydrogen only. The molecular formula for hydrocarbon compounds is CxHy. There are different types of hydrocarbons such as saturated hydrocarbons, unsaturated hydrocarbons, cycloalkanes, aromatic hydrocarbons and aliphatic hydrocarbons.
What are the two main classes of hydrocarbons?
There are many hydrocarbons. They can be classified into two main classes: aliphatic and the aromatic hydrocarbons. Aliphatic hydrocarbons are composed of catenated carbon chain. In acyclic compounds, the carbon chain are straight or branched.
What determines the chemical and physical properties of hydrocarbons quizlet?
What determines the chemical and physical properties of hydrocarbons? The way the atoms are connected together, and the number of carbon and hydrogens (both A and B)
What is the definition of a hydrocarbon 1 point quizlet?
Hydrocarbons. The simplest organic compounds. It is a compound that only contains carbon and hydrogen atoms.
Which compound contains nitrogen?
FractionCompoundsUse by MicroorganismsInorganic nitrogenAmmonia and ammonium: NH3 and NH4+Easily assimilatedOrganic nitrogenAmino acids with a molecular mass of <200 DaPolypeptides with a molecular mass of 200–10,000 Da.Non-assimilableProteins with a molecular mass of >10,000 Da
What are hydrocarbons class 10th?
Hydrocarbon. Hydrocarbon. All carbon compounds having carbon and hydrogen are known as hydrocarbons. The saturated hydrocarbons having single bonds are called alkanes. For example, Methane, Ethane etc.