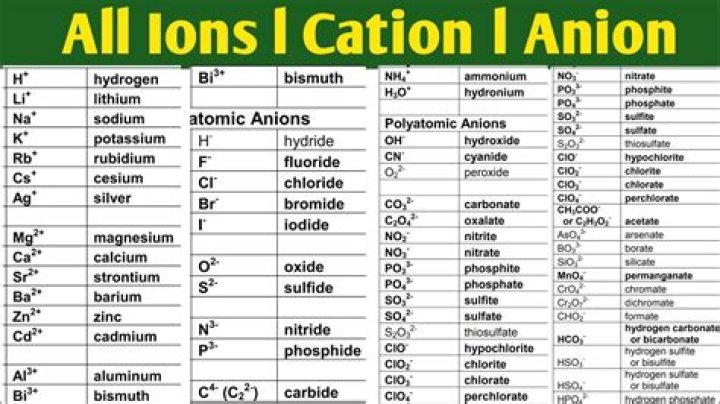
What About Common Ions? What Are Those?
- Common Simple Cations: aluminum Al3+, calcium CA2+, copper Cu2+, hydrogen H+, ferrous iron Fe2+, ferric iron Fe3+, magnesium Hg2+, mercury (II) Mg2+, potassium K+, silver Ag+, Sodium Na+.
- Common Simple Anions: chloride C–, fluoride F–, bromide Br–, oxide O2-
.
Considering this, what are the 4 most common ions?
The fours most abundant ions in the body are potassium, sodium, calcium, and chloride.
One may also ask, what is the most common ion for oxygen? Oxygen, O. Oxygen is in Group 6. It has six electrons in its outer shell. It gains two electrons from one or two other atoms in reactions, forming an oxide ion, O 2-.
Also to know, what are the common ions?
- lead (IV) ion.
- tin (IV) ion.
- mercury (I) ion*
- manganese (II) ion.
- cobalt (III) ion.
- copper (II) ion.
- iron (III) ion.
- manganese (III) ion.
Is HCl a cation or anion?
HCl, which contains the anion chloride, is called hydrochloric acid. HCN, which contains the anion cyanide, is called hydrocyanic acid. Rules for Naming Oxyacids (anion contains the element oxygen): Since all these acids have the same cation, H+, we don't need to name the cation.
Related Question AnswersDo we have ions in our body?
Body fluid contains electrolytes, chemicals which, when they dissolve in water, produce charged ions. These ions enable the flow of electrical signals through the body. Electrolytes play an important role in the body; they regulate the osmotic pressure in cells and help maintain the function of muscle and nerve cells.How many ions are in the human body?
Roles of Electrolytes. These six ions aid in nerve excitability, endocrine secretion, membrane permeability, buffering body fluids, and controlling the movement of fluids between compartments. These ions enter the body through the digestive tract.Is oxygen a cation or anion?
By itself, oxygen is neutral (does not have a charge). In order to be considered a cation or anion, there must be charge involved. Although it actually depends on whether the anode/cathode is considered positive or negative (both can be either, although the other must be the opposite).What is ions in biology?
An ion is defined as an atom or molecule that has gained or lost one or more of its valence electrons, giving it a net positive or negative electrical charge. In other words, there is an imbalance in the number of protons (positively charged particles) and electrons (negatively charged particles) in a chemical species.What is the most common ion of calcium?
The calcium ion is also known as Ca2+, meaning it has two less electrons than protons and is not stable in nature.Why do ions form?
Ions are formed when atoms lose or gain electrons in order to fulfill the octet rule and have full outer valence electron shells. When they lose electrons, they become positively charged and are named cations. When they gain electrons, they are negatively charged and are named anions.How do you name ions?
Method 1 Naming a Monoatomic Ion With a Single Oxidation State- Memorize the periodic table. In order to remember the names of ions, you will need to remember the names of the elements that form them.
- Remember to add the word ion.
- Use element names for positive ions.
- Add the -ide suffix for negative ions.
What are examples of polyatomic ions?
Indeed, most ionic compounds contain polyatomic ions. Well-known examples are sodium hydroxide (NaOH) with OH- as the polyatomic anion, calcium carbonate (CaCO3), and ammonium nitrate (NH4NO3), which contains two polyatomic ions: NH+ and NO3-.Is oxygen a polyatomic ion?
Polyatomic Ions. A polyatomic ion is an ion composed of more than one atom. The ammonium ion consists of one nitrogen atom and four hydrogen atoms. The hydroxide ion (OH-) is an oxygen atom (red) bonded to a hydrogen atom.Is indium a cation or anion?
Most Group 3 metals (aluminum, gallium, and indium) form 3+ cations. The cation of aluminum, thus, is designated as Al 3+ . Group 6 nonmetals and metalloids (oxygen, sulfur, selenium, and tellurium) form 2− anions.What is potassium's most common ion?
Potassium- MELTING POINT: 63.38°C.
- BOILING POINT: 759°C.
- DENSITY: 0.862 g/cm 3
- MOST COMMON IONS: K +