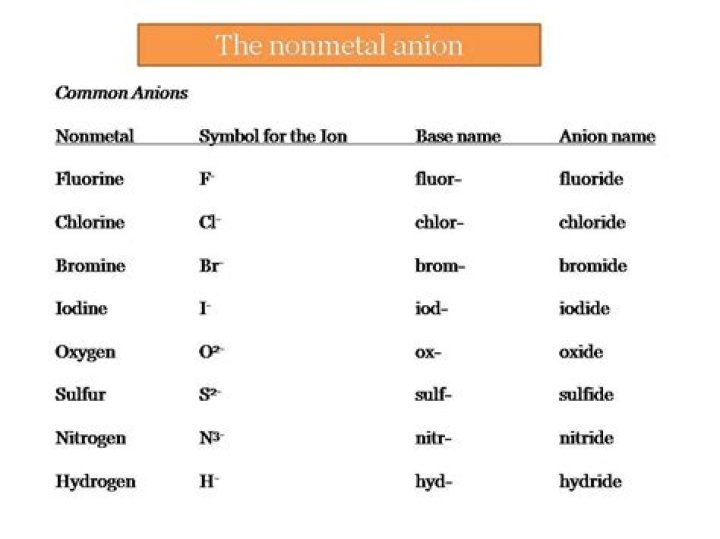
- Bromide - Br -
- Chloride - Cl -
- Fluoride - F -
- Iodide - I -
- Nitride - N 3-
- Oxide - O 2-
- Sulfide - S 2-
.
In this manner, what are common anions?
Common anions: chloride, fluoride, bromide, nitrate, nitrite, sulfate, and phosphate. Inorganic disinfection byproducts (DBPs): chlorate, chlorite, and bromate. Other anions, such as perchlorate and chromate.
Furthermore, what are anions and cations give examples? Some examples of anions are Iodide (I–), chlorine (Cl–), hydroxide (OH–). When sodium a cation is depicted as NA+, the plus charge indicator shows that it has one electron less than the total number of protons. Thus sodium having an uneven distribution of electrons and protons enables it to have a positive charge.
Then, what are examples of cations?
Examples of Cations For example, sodium always forms a +1 cation and magnesium always forms a +2 cation. Some metals are flexible and can form more than one type of cation. Copper can form +1 or +2 cations, and iron can form +2 or +3 cations.
What type of elements form anions?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).
Related Question AnswersIs HCl an anion?
HCl, which contains the anion chloride, is called hydrochloric acid. HCN, which contains the anion cyanide, is called hydrocyanic acid. Rules for Naming Oxyacids (anion contains the element oxygen): Since all these acids have the same cation, H+, we don't need to name the cation.Is h2o a cation or anion?
Ions with a positive charge are called "Cations" and ions with a negative charge are called "Anions". Ion exchange resins are used to exchange non desirable cations and anions with hydrogen and hydroxyl, respectively, forming pure water (H20), which is not an ion.Is NaCl a cation or anion?
Sodium chloride is written NaCl, where Na+ is the cation and Cl- is the anion. An anion is an ionic species having a negative charge.How are anions formed?
Positive and Negative Ions: Cations and Anions. Cations (positively-charged ions) and anions (negatively-charged ions) are formed when a metal loses electrons, and a nonmetal gains those electrons. And all of them form an anion with a single negative charge.Is cyanide an anion or cation?
The term cyanide refers to a singularly charged anion consisting of one carbon atom and one nitrogen atom joined with a triple bond, CN-. The most toxic form of cyanide is free cyanide, which includes the cyanide anion itself and hydrogen cyanide, HCN, either in a gaseous or aqueous state.Which is an example of an anion?
For example consider sodium cations (Na+) and Chlorine anions (Cl-). Sodium has a positive 1 charge and chloride has a negative 1 charge. Thus one sodium cation cancels one chloride anion resulting in the formula Na1Cl1 or NaCl.Is silver a cation or anion?
Aluminum and the elements in group 3 are always +3 when they form cations. Zinc and cadmium always form +2 cations. Although silver can form both +1 and +2 cations, the +2 is so rare that we usually name Ag+ as silver ion, not silver(I) ion. Ag2+ is named silver(II) ion.What are basic cations?
The main ions associated with CEC in soils are the exchangeable cations calcium (Ca2+), magnesium (Mg2+), sodium (Na+) and potassium (K+) (Rayment and Higginson 1992), and are generally referred to as the base cations. In most cases, summing the analysed base cations gives an adequate measure of CEC ('CEC by bases').What are common cations?
Terms in this set (48)- Hydrogen ion. H?
- Lithium ion. Li?
- Sodium ion. Na?
- Potassium ion. K?
- Cesium ion. Cs?
- Silver ion. Ag?
- Magnesium ion. Mg²?
- Calcium ion. Ca²?
How do you know if an element is a cation or anion?
Key Points- The electronic configuration of many ions is that of the closest noble gas to them in the periodic table.
- An anion is an ion that has gained one or more electrons, acquiring a negative charge.
- A cation is an ion that has lost one or more electrons, gaining a positive charge.
Is Iron positive or negative?
A: The iron(III) ion has a charge of +3, so it has one less electron than the iron(II) ion, which has a charge of +2.Names and Symbols.
| Cations | Iron(III) ion |
|---|---|
| Fe3+ | |
| Anions | Oxide |
| O2- |