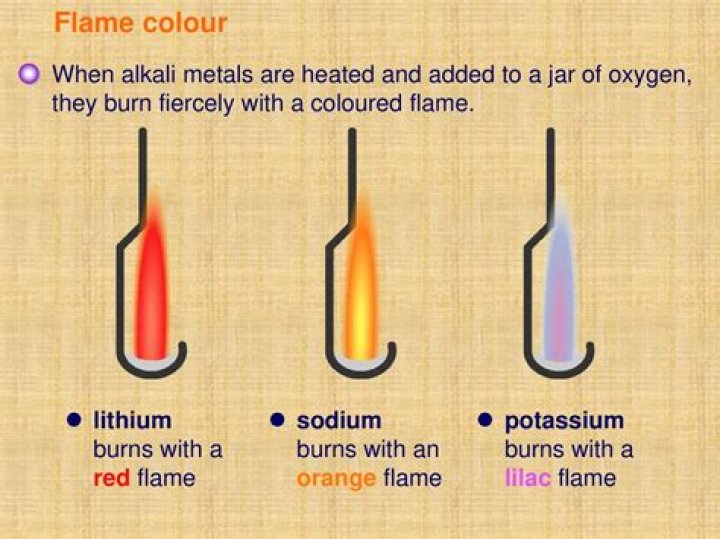
.
Regarding this, what is produced in burning?
Products of combustion. When a compound burns it reacts with oxygen. The oxygen will combine with the carbon and the hydrogen in the methane molecule to produce carbon dioxide (CO 2) and water (H 2O).
Beside above, what causes a substance to form a flame? Flames are the result of a chemical reaction, primarily between oxygen in the air and a fuel, such as wood or propane. In addition to other products, the reaction produces carbon dioxide, steam, light, and heat. If the flame is hot enough, the gases are ionized and become yet another state of matter: plasma.
Herein, how do substances burn with or without a flame?
The chemical reaction is going on in the substance being burned, and in the air above it as the heat of combustion vaporizes some of the substance being burned. If your reaction does not have a flame, then either it is not producing visible light or the reaction does not occur in the air above the substance.
Why does a matchstick produce a flame on burning?
A matchstick starts burning on rubbing it on the side of the matchbox because the heat produced by friction heats the chemical at the head of the matchstick to their ignition temperature and makes it catch fire.
Related Question AnswersIs fire hot or cold?
There is a type flame called “cool” fire, but that is considered to be below 400° C or 752° F. This type of fire is chemically induced, and emits very little heat. But it is still hot enough to burn you. If you consider that a nice campfire hits about 1200°-1400° F, half that temperature is still enough to burn you.What is perfect combustion?
Perfect combustion is simply a mixture of fuel and oxygen, with both being completely consumed in the burning process. The ideal situation would be to provide just enough air in the combustion chamber to insure complete burning of the fuel.What is the formula for combustion?
A combustion reaction occurs when a substance reacts quickly with oxygen (O2). The general equation for a complete combustion reaction is: Fuel + O2 → CO2 + H2O. The burning of charcoal is a combustion reaction.Does water help combustion?
Short answer: Water is formed as a result of the combustion of hydrogen. In simple words, water is what you get when you burn hydrogen. So, water doesn't burn because, in a way, it has already burned.What are the four products of combustion?
Products of Combustion- Carbon Dioxide.
- Carbon Monoxide.
- Sulfur Dioxide.
- Nitrogen Oxides.
- Lead.
- Particulate Matter.
What are the 3 types of combustion?
The three important types of combustion are:- Rapid combustion.
- Spontaneous combustion.
- Explosive combustion.
What are the three elements of fire?
The triangle illustrates the three elements a fire needs to ignite: heat, fuel, and an oxidizing agent (usually oxygen).Is fire a plasma?
Fire (flames) may contain plasma, albeit one that is a partially ionized plasma, and dominated by collisions: “Whether a plasma exists in a flame depends on the material being burned and the temperature”. fire) is shown as a plasma.What are the conditions for combustion?
Three things are required in proper combination before ignition and combustion can take place---Heat, Oxygen and Fuel.- There must be Fuel to burn.
- There must be Air to supply oxygen.
- There must be Heat (ignition temperature) to start and continue the combustion process.