A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge..
Hereof, is water polar or nonpolar?
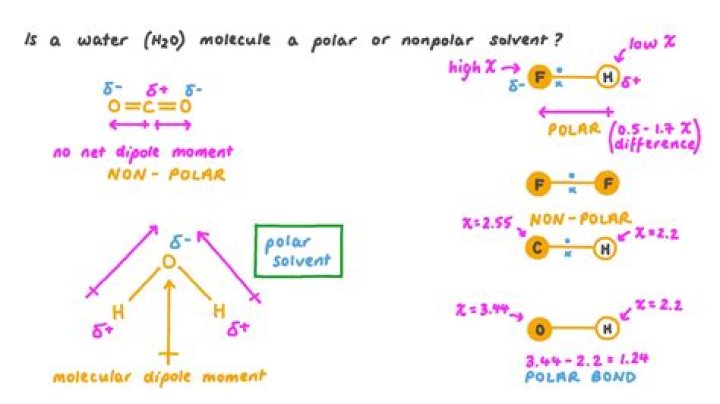
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5.
Beside above, is HCL polar or nonpolar? HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?
Similarly, it is asked, what type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.
Why Water is a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.
Related Question Answers
What is Polar vs nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.Is NaCl a polar molecule?
Yes, NaCl is an ionic bond which makes it polar. A polar molecule consists of atoms having either a positive or negative bond. In this case, Na has a +1 charge and Cl has a -1 charge, making the bond polar. The difference in electronegativities is what makes a bond polar or nonpolar.Is co2 a polar molecule?
In general, a bond between two atoms with an χ difference of 0.4 to 1.7 (on the Pauling scale) is considered polar covalent. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar.Is benzene polar or nonpolar?
We could say that benzene is nonpolar because it is a hydrocarbon that contains only C-C and C-H bonds, and hydrocarbons are nonpolar. But C is slightly more electronegative than H (by 0.35 units), so a C-H bond is very slightly polar and has a small dipole moment.Is Sugar polar or nonpolar?
Table sugar (sucrose) is a polar nonelectrolyte. Sucrose is quite soluble because its molecules bristle with water-accessible OH groups, which can form strong hydrogen bonds with water. So sugar is not an exception to the "like dissolves like" rule of thumb. Nonelectrolytes can be either polar or nonpolar.Is HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.Is h2o covalent bond?
In H2O molecule, two water molecules are bonded by a Hydrogen bond but the bond between two H - O bonds within a water molecule are covalent. The dotted lines represent a hydrogen bond and the solid lines represent a covalent bond.Why is water a polar covalent bond?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Is water an ionic bond?
Water (H2O) can be called a molecule or a compound because it is made of hydrogen (H) and oxygen (O) atoms. There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Ionic bonds occur when electrons are donated from one atom to another.Is NaCl a covalent bond?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.How is a covalent bond formed?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.What type of bond is Salt?
Salt is made up of sodium and chloride and is ionically bonded. Sugar, on the other hand, is composed of carbon, oxygen, and hydrogen and has covalent bonds. A salt molecule is made up of one sodium atom and one chlorine atom. For salt to be made, the sodium atom must lose an electron and become a sodium ion.What kind of bond is NaCl?
Salt or Sodium Chloride (NaCl) is a good example of a ionic bonding. Sodium (Na) has 1 valance electron and Chlorine (Cl) has 7 electrons in its outer orbit. If Sodium lost its valance electron, its next shell will be full. But that would also make Sodium a positive ion.Is water a single covalent bond?
Water is not a covalent bond. Rather, the bonds in H2O are covalent. And so, H2O is a simple covalent molecule. The oxygen atom shares an outermost electron each with one hydrogen atom (which also shares an outermost electron) to give a single bond.Is CO A polar covalent bond?
Carbon monoxide is a hetero nuclear diatomic molecule. It is a polar covalent molecule as the electronegativity difference of oxygen and carbon is greater than 0.4, hence, forms a polar covalent bond.Is HCl a polar covalent bond?
Hydrogen has an electronegativity of 2.1, and chlorine has an electronegativity of 3.0. The electron pair that is bonding HCl together shifts toward the chlorine atom because it has a larger electronegativity value. A bond in which the electron pair is shifted toward one atom is called a polar covalent bond.Is cl2 polar or nonpolar?
2. Why is an HCl molecule polar while a Cl2 molecule is nonpolar? H & Cl have different electronegativities; therefore they form a polar bond. Cl & Cl have same electronegativity; therefore they form a nonpolar bond.Is bcl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.What happens if a covalent bond is polar?
In a polar covalent bond, the electrons shared by the atoms spend a greater amount of time, on the average, closer to the Oxygen nucleus than the Hydrogen nucleus. This is because of the geometry of the molecule and the great electronegativity difference between the Hydrogen atom and the Oxygen atom.