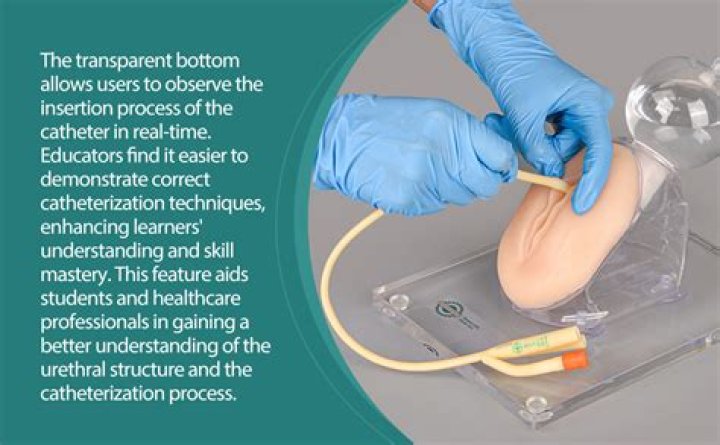
Ions -- electrically charged atoms -- can carry a positive or negative charge. Positive ions are cations and are typically metals like copper or sodium. Negatively-charged ions are anions, formed from nonmetallic elements like oxygen and sulfur..
Also, is Metal positively or negatively charged?
Metal atoms lose the electron, or electrons, in their highest energy level and become positively charged ions. Non-metal atoms gain an electron, or electrons, to become negatively charged ions.
Also, can metal be charged? Metals can be charged if sufficient electrons can be removed through friction but there will still be no localisation of charge. While in case of insulators when electrons are removed or added through friction they can't get redistributed as electrons are not free ,thus making them charged.
are metals always positive?
Notice that all metals lose electrons to form positive ions. Group 1 metals commonly lose only one electron to form ions with a single positive charge. Other metals, like the transition metals, also always form positive ions, but the number of electrons they may lose varies. Nonmetals form ions by gaining electrons.
Is Iron negatively charged?
Since Iron, Fe, is a transition metal it usually will conform it's positive charge to an element or compound that already has a set negative charge for example iron is a transition metal and because its orbitals are so close energy wise they tend to give up either 2 or 3 electrons at time.
Related Question Answers
Is oxygen positive or negative?
A good example is an oxygen atom that is sharing one electron with each of two nearby hydrogen atoms, making a molecule of water H2O. An oxygen atom that has successfully donated one or two electrons or is sharing one or two electrons has lost part of its negative charge, so it is left positively charged.Why is a metal positively charged?
Metal elements form positively charged ions called cations because they are located on the left side of the periodic table. These elements all have valence electrons in an s orbital. These electrons are relatively easy for the atom to lose to achieve a stable octet of electrons in its outermost energy shell.Is hydrogen positive or negative?
One Hydrogen (H) has a positive charge +1. This has nothing to do with it having more protons than electrons, as it has one of each. Think of it as 'willing' to donate one electron, or it has +1 electrons see, it is on the plus side. One Oxygen (O) has a negative charge of -2.Are metals malleable?
The energy is transferred throughout the rest of the metal by the moving electrons. Metals are described as malleable (can be beaten into sheets) and ductile (can be pulled out into wires). This is because of the ability of the atoms to roll over each other into new positions without breaking the metallic bond.Is aluminum positive or negative?
Aluminum, a member of the IIIA family, loses three electrons to form a 3+ cation. The halogens (VIIA elements) all have seven valence electrons. All the halogens gain a single electron to fill their valence energy level. And all of them form an anion with a single negative charge.Are all metals cations?
Metals are not cations, metal ions are. Any substance in elemental form is an atom or a molecule. They become ions depending upon the the electronic configuration. Positive ions(cations) is they lose electrons or negative ions(anions).Is lead positive or negative?
The black (common) lead on the multimeter indicates the negative (cathode) lead, and the red indicates the positive or anode side.Are cations always Metals?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).Are all non metals negatively charged?
The outer shells of non-metal atoms gain electrons when they form ions: the ions formed are negative, because they have more electrons than protons. the ions have the electronic structure of a noble gas (group 0 element), with a full outer shell.Do metals always lose electrons?
Metals tend to lose electrons and non-metals tend to gain electrons, so in reactions involving these two groups, there is electron transfer from the metal to the non-metal. The metal is oxidized and the non-metal is reduced.Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.How do you know if an element is neutral?
That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. Ions are atoms with extra electrons or missing electrons. When you are missing an electron or two, you have a positive charge. When you have an extra electron or two, you have a negative charge.How do u know if an element is positive or negative?
To find the ionic charge of an element you'll need to consult your Periodic Table. On the Periodic Table metals (found on the left of the table) will be positive. Non-metals (found on the right) will be negative.Is magnesium positive or negative?
A positive sign is added to the symbol for sodium, Na +. Ions with greater charge include a number in their symbol, for example Al 3+ (which has three positive charges). Magnesium is in group 2. How many electrons are in the outer shell of a magnesium atom?Is Salt positively or negatively charged?
For example, in the compound sodium chloride — table salt — the sodium atom has a positive charge and the chlorine atom has a negative charge. This unequal number of negative and positive charges can occur in one of two ways: An atom can gain a proton (a positive charge) or lose an electron (a negative charge).What is a positive ion called?
An ion is an atom or group of atoms where the number of electrons is not equal to the number of protons. When an atom gains electrons, this results in a negative charge. This type of ion is called an anion. When an atom loses electrons, this results in a positive charge. A positively charged ion is called a cation.What is positive and negative ions?
To put it simply, positive ions are molecules that have lost one or more electrons whereas negative ions are actually oxygen atoms with extra-negatively-charged electrons. Negative ions are abundant in nature, especially around waterfalls, on the ocean surf, at the beach and after a storm.What is charge made of?
What Is Electric Charge? Protons and electrons create electric fields. Most electric charge is carried by the electrons and protons within an atom. Electrons are said to carry negative charge, while protons are said to carry positive charge, although these labels are completely arbitrary (more on that later).How is charge created?
Electric charge is carried by subatomic particles. In ordinary matter, negative charge is carried by electrons, and positive charge is carried by the protons in the nuclei of atoms. An electric charge has an electric field, and if the charge is moving it also generates a magnetic field.