.
Also, how many filled Subshells does arsenic have?
Arsenic is found in the fourth period of the table of elements. It is a member of the phosphorus family with other elements including phosphorus (duh), antimony (Sb), and bismuth (Bi). All of the members of this family have five electrons in their outer orbital.
Similarly, how many neutrons does arsenic have? 42 neutrons
Keeping this in view, how many energy shells does arsenic have?
A natural chemical An arsenic atom has 33 electrons and 33 protons with five valence electrons (those that can participate in forming chemical bonds with other electrons) in its outer shell.
Why is 3rd shell 8 or 18?
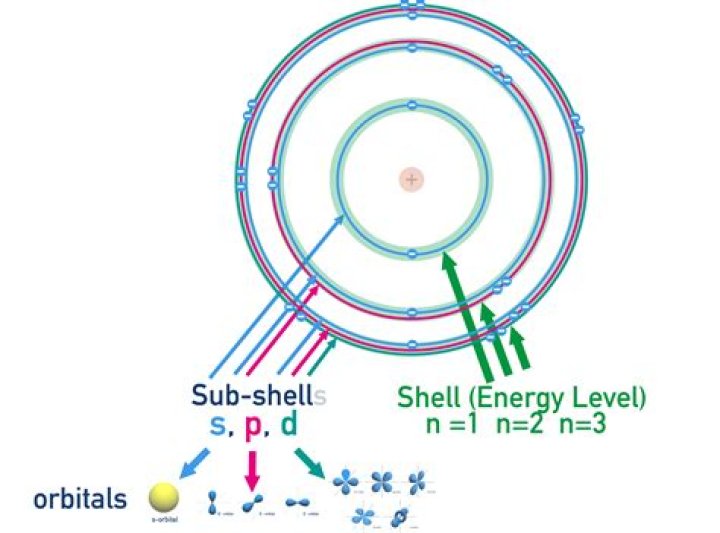
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. For an explanation of why electrons exist in these shells see electron configuration.
Related Question AnswersWhat color is arsenic?
grayWhat is the family name of arsenic?
Arsenic, atomic number 33 lies in between phosphorus and antimony in group 15, the so called Nitrogen group of the periodic table.What Subshells are allowed in the N 3 shell?
It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The n = 3 shell, for example, contains three subshells: the 3s, 3p, and 3d orbitals. There is only one orbital in the n = 1 shell because there is only one way in which a sphere can be oriented in space.What does arsenic react with?
When heated in oxygen, arsenic ignites to form "arsenic pentoxide" - actually tetra-arsenic decaoxide. Arsenic reacts with fluorine to form the gas arsenic(V) fluoride. Arsenic reacts under controlled conditions with the halogens fluorine, chlorine, bromine and iodine to form arsenic(III) trihalides.What is the electron configuration of mg2+?
Therefore the Magnesium electron configuration will be 1s22s22p63s2. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom.Is arsenic reactive?
Arsenic is a metalloid element, which, like phosphorus, has two main forms. One is a brittle, gray metallic form and the other is a yellow, non-metallic solid. Yellow arsenic is the more reactive and unstable of the pair. Arsenic is stable, being unaffected by air, water, most acids and alkalis.What is arsenic made from?
Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. Arsenic and its compounds, especially the trioxide, are used in the production of pesticides, treated wood products, herbicides, and insecticides.What is arsenic electron configuration?
[Ar] 3d¹° 4s² 4p³Where is arsenic most commonly found?
2.1 Arsenic is found in the natural environment in some abundance in the Earth's crust and in small quantities in rock, soil, water and air. It is present in many different minerals. About one third of the arsenic in the atmosphere comes from natural sources, such as volcanoes, and the rest comes from man-made sources.What energy level is arsenic in?
| Number of Energy Levels: | 4 |
|---|---|
| First Energy Level: | 2 |
| Second Energy Level: | 8 |
| Third Energy Level: | 18 |
| Fourth Energy Level: | 5 |