Questions and Answers
| Energy Level (Principal Quantum Number) | Shell Letter | Electron Capacity |
|---|---|---|
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
.
Similarly, how many orbitals does n 4 have?
16 orbitals
Subsequently, question is, how many electrons can be contained in all of the orbitals with N 2? (a) When n = 2, there are four orbitals (a single 2s orbital, and three orbitals labeled 2p). These four orbitals can contain eight electrons. Again, each orbital holds two electrons, so 50 electrons can fit in this shell.
In respect to this, what is the maximum number of electrons possible with N 4 in an atom?
The maximum number of electrons will be if the 4th shell is completely filled. But if the max possible n = 4, then 36 electrons is the answer (18 + 18 for 18 groups).
How many electrons are there in a filled N 4 principal shell?
32 electrons
Related Question AnswersHow many Subshells does n 4 have?
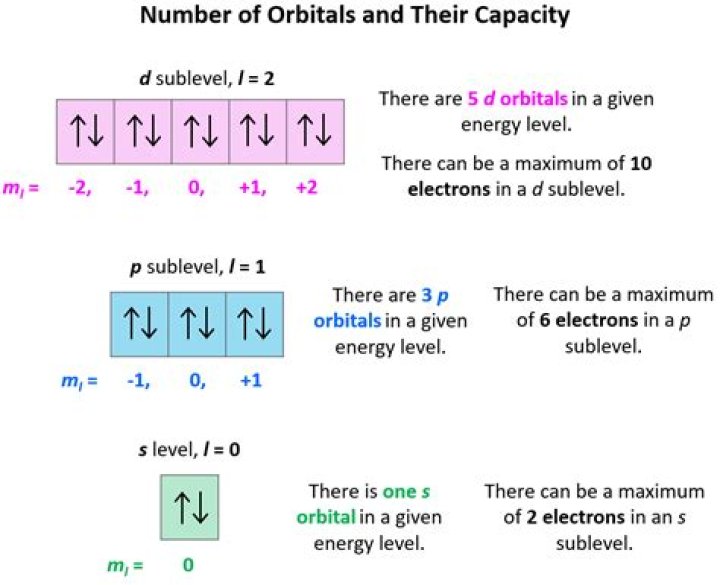
The fourth shell has 4 subshells: the s subshell, which has 1 orbital with 2 electrons, the p subshell, which has 3 orbitals with 6 electrons, the d subshell, which has 5 orbitals with 10 electrons, and the f subshell, which has 7 orbitals with 14 electrons, for a total of 16 orbitals and 32 electrons.What Subshell is N 5?
For n = 5, the possible values of l = 0,1,2,3,4. These numbers correspond to s, p, d, f and g orbitals. Now, s has 1 subshell, p has 3, d has 5, f has 7 and g has 9. Thus, total number of subshells = 25.How many orbitals are in 1s?
One spin-up and one spin-down. This means that the 1s, 2s, 3s, 4s, etc., can each hold two electrons because they each have only one orbital. The 2p, 3p, 4p, etc., can each hold six electrons because they each have three orbitals, that can hold two electrons each (3*2=6).How many orbitals are in 5f?
sevenWhat is a Subshell?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.How many orbitals are in 2s?
one orbitalHow many Subshells are in the N 4 principal shell?
Re: How many subshells there are in the shell n=4? Answer: There are 4 subshells: 4s, 4p, 4d, and 4f.How many types of orbitals are there in the shell with N 4 in an atom?
and each ml value corresponds to one orbital. We have 4− subshells in this case; s,p,d,f ↔ 0,1,2,3 for the value of l . =16−− orbitals in the n=4− energy level. If you repeat the process for n=3 , you would find lmax=2 and there are 9− orbitals in n=3− .How many electrons are in a ml 0?
Table of Allowed Quantum Numbers| n | l | Number of electrons |
|---|---|---|
| 1 | 0 | 2 |
| 2 | 0 | 2 |
| 1 | 6 | |
| 3 | 0 | 2 |