The slideshow shows a
covalent bond being formed between a hydrogen
atom and a
chlorine atom, to
form hydrogen chloride. After
bonding, the
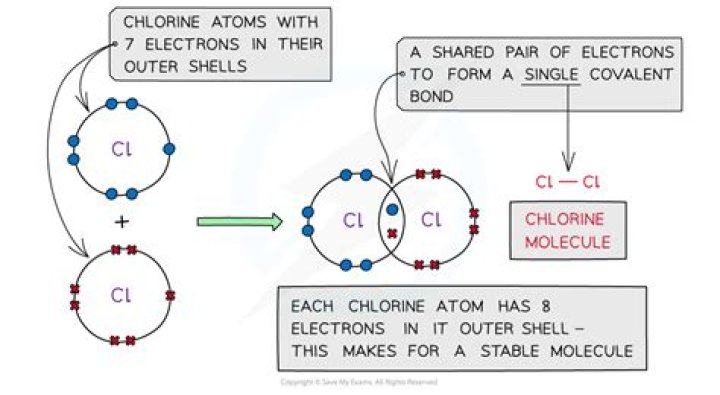
chlorine atom is now in contact with eight electrons in its outer shell, so it is stable .
How many bonds?
| Element | Number of bonds |
| Group 7 | Chlorine | 8 - 7 = 1 |
.
Likewise, how many covalent bonds can chlorine form?
Double and Triple Bonds[edit] Covalent bonds can also form between other non-metals, for example chlorine. A chlorine atom has 7 electrons in its valence shell—it needs 8 to complete it. Two chlorine atoms can share 1 electron each to form a single covalent bond. They become a Cl2 molecule.
Furthermore, what element does chlorine form a covalent bond with? Hydrogen on sharing its one electron with chlorine results in formation of hydrogen chloride molecule and chlorine and hydrogen complete its octet by sharing one electron of each and thus forming a single covalent bond by sharing of two electrons. Thus, Hydrogen is the element that forms covalent bond with chlorine.
Keeping this in view, how many bonds can an atom form?
The number of electrons required will be the number of bonds formed most of the time. A hydrogen atom can form 1 bond, a carbon atom can form 4 bonds, a nitrogen atom can form 3 bonds and an oxygen atom can form 2 bonds. H Atom- 1s1, it can only form 1 covalent bond.
How covalent bond is formed?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.
Related Question Answers
Are covalent bonds strong?
Covalent bonds between atoms are quite strong, but attractions between molecules/compounds, or intermolecular forces, can be relatively weak. Covalent compounds generally have low boiling and melting points, and are found in all three physical states at room temperature.What are examples of covalent bonds?
Examples of Covalent Bond: - Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.
How many electrons does a covalent bond consist of?
two electrons
What are the three types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.How many covalent bonds are formed by F?
So fluorine and bromine are halogens (Group VII) and when you work out the Lewis dot structure you'll see that they each have one unpaired electron, therefore these unpaired electrons join together to form one bond. Oxygen with two unpaired electrons forms two bonds as a diatomic molecule and typically in compounds.Can oxygen form a triple bond?
If oxygen starts with 6 electrons and forms a triple bond, then it has 2 lone pair electrons. Using the formal charge formula, 6 - (2+6/2) = 1. Since oxygen is so electronegative, it'll tend not to have a positive formal charge. However, oxygen can form triple bonds with carbon and nitrogen.Why do hydrogen and chlorine form a single covalent bond?
chlorine and hydrogen atoms form a single covalent bond. in hydrogen chloride gas because the electrons are shared, not transferred from one atom to another. when it is dissolved in water to become hydrochloric acid.What happens when covalent bonds break?
Covalent bonds form between atoms when the total energy present in the newly formed molecule is lower than the energy present in each of the atoms alone. Breaking covalent bonds requires energy, and covalent bond formation releases energy. The term used to describe the energy in a system is Gibbs Free Energy.Is Co a single covalent bond?
In these examples, each individual atom has just one unpaired electron to share with the other atom. When a covalent bond forms with one pair of shared electrons, we call it a single covalent bond. Methane, which is a carbon atom with single covalent bonds to four hydrogen atoms, is the simplest carbon compound.How many electrons are in a triple bond?
One covalent bond contains 2 electrons, one from each atom. A triple bond indicates that, well, there are three total covalent bonds in that one bond. So, 3 bonds x 2 electrons per bond would give you 6 total electrons.Can nitrogen have 4 bonds?
If you look at the above image you can see that when nitrogen has a positive charge (one less electron), it can form four covalent bonds. Either with single, double, or triple bonds. It is similar to phosphorus in this regard because they both have five valence electrons (four when they have a positive charge).What is a polar covalent bond?
Nonpolar covalent bonds are a type of chemical bond where two atoms share a pair of electrons with each other. Polar covalent bonding is a type of chemical bond where a pair of electrons is unequally shared between two atoms.How many electrons are shared in a single covalent bond?
two electrons
Why is oxygen a diatomic molecule?
Oxygen generally exists as a diatomic molecule in the atmosphere when it is not combined with any other element. It forms the molecule O2 because in that configuration, it has its lowest energy level when uncombined. All substances tend to go to the lowest energy level possible. Oxygen has 6 valence electrons.What type of bond is o2?
covalent bonds
Can oxygen have 4 bonds?
Theoretically, yes. By looking at the electronic configuration you can derive the formal charge. Oxygen with no bonds has 6 electrons in the outer shell, and it would like to have eight. Oxygen with four bonds would have four electrons from neighbors, so to get to 8 it needs to shed two of its own six.How many lone pairs does hydrogen have?
Hydrogen atoms have 1 bond and no lone pairs. Oxygen atoms usually have 2 bonds and 2 lone pairs. Hydrogen atoms have 1 bond and no lone pairs.Is n2 a covalent bond?
Triple covalent bond is present in nitrogen. Nitrogen is a non metal. A nitrogen atom has 5 electrons in its outer shell. Two nitrogen atoms will each share three electrons to form three covalent bonds and make a nitrogen molecule (N2).Which pair of elements will form a covalent bond?
(A) Both hydrogen and chlorine are non-metals and the bond formed between two non-metals is a covalent bond. So hydrogen and chlorine will form covalent bonds with each other and the resulting compound HCl will be covalent compound. (B) Sodium is metal while chlorine is a non-metal.