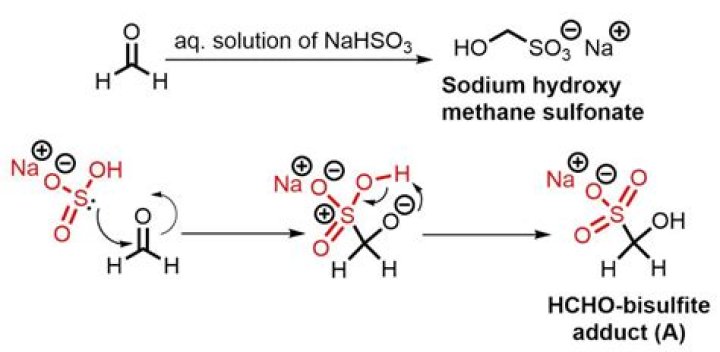
Add 4.75 g of sodium metabisulfite (di-sodium disulfite, Na2S2O5) to 6.25 mL of sterile, degassed ddH2O.Add 1.75 mL of 2 m NaOH.Add 1.25 mL of 1 m hydroquinone (0.11 g in 1 mL of H2O).Heat to 50°C in the dark, inverting the tube frequently.Adjust the pH to 5.0.
How do you make sodium bisulfate?
Production. Sodium bisulfate is produced as an intermediate in the Mannheim process, an industrial process involving the reaction of sodium chloride and sulfuric acid: NaCl + H2SO4 → HCl + NaHSO. This step is highly exothermic.
What is the difference between sodium bisulfate and sodium bisulfite?
In inorganic chemistry|lang=en terms the difference between bisulfite and bisulfate. is that bisulfite is (inorganic chemistry) the univalent group -hso3, or any salt containing it while bisulfate is (inorganic chemistry) any salt containing this anion.
What is sodium bisulfite solution?
Sodium bisulfite solution is a variously colored liquid at room temperature. It is used in waste water treatment, in the pulp and paper industry, in the photographic industry and in various other industries as a bleach or dechlorinator. It can also be used to treat flue gases to remove sulfur trioxide (SO3).What does sodium bisulfite do to water?
Sodium Bisulfite is primarily used for water treatment to remove excess chlorine in drinking water.
Is sodium a bisulfite?
Sodium Bisulfite is a white, crystalline solid with a slight odor of rotten eggs. It is often in a liquid solution. It is used in making paper and leather, as a food preservative and in dye and chemical production. Substance List because it is cited by ACGIH, DOT, NIOSH, IARC and EPA.
Is baking soda sodium bisulfate?
NamesRelated compoundsSodium bisulfate Sodium hydrogen phosphate
Are sodium bisulfite and sodium metabisulfite the same thing?
The key difference between sodium bisulfite and sodium metabisulfite is that the sodium bisulfite has only one sulfur atom and three oxygens, and the bisulfite anion is monovalent whereas, the sodium metabisulfite has two sulfur atoms, five oxygens, and the anion is divalent.How is sodium bisulfite manufactured?
Sodium bisulfite, NaHSO 3, can be prepared by reacting sulfur dioxide gas, SO 2, in a solution containing alkaline hydroxide, e.g. sodium hydroxide (NaOH) or sodium carbonate (Na 2CO 3). … Reacting a solution of sodium sulfite with additional sulfur dioxide gas can produce sodium bisulfite.
What contains sodium bisulfite?Sodium bisulfate is often used in toilet bowl cleaners and dishwasher cleaning products. It can also be found in a multitude of other items. Commonly used to control pH, it is found in aquarium and swimming pool products. It can even be found in dietary supplements and antifungal foot creams.
Article first time published onHow do you neutralize sodium bisulfite?
Any spillages should be neutralized with soda ash, to prevent sulphur dioxide emission, and then be oxidized to neutral sulphate with sodium hypochlorite. Sodium metabisulphite should be used if dechlorinating chemical is required in powder form.
Is sodium bisulfite edible?
Is Sodium Metabisulfite Safe to Eat? Yes, it almost has no side effects and the safety has been approved by the U.S. Food and Drug Administration (FDA) and European Food Safety Authority (EFSA), as well as the Joint FAO/WHO Expert Committee on Food Additives (JECFA).
How do I add sodium bisulfate to my pool?
- Turn the jets off if you have an inground pool.
- Add the sodium bisulfate slowly so as not to disrupt the pH balance.
- Wait another 6 hours before you retest.
How do you make sodium bicarbonate solution?
Sodium bicarbonate (baking soda) is only mildly soluble in water, and can only dissolve 8 grams of it in 100mL of water at room Temp. It increases in solubility as the temperature goes higher. If you need to dissolve large amounts, just add a lot of water, and then heat it up.
Is Arm and Hammer baking soda the same as sodium bicarbonate?
Baking soda is Sodium Bicarbonate. Whether produced by the Arm and Hammer brand or a generic or ANY other brand name it is STILL just 100% pure sodium bicarbonate. So unless having a particular logo and box shade matters to you, there is ZERO reason to pay more for any brand name baking soda. here is NO difference.
How soluble is sodium bisulfite?
Sodium Bisulfite is a preservative that exists as a powder, with a solubility of 1 g in 4 ml of water.
What is sulfite sodium bisulfite?
Sodium bisulfite (SBS), also known as sodium bisulphite or sodium hydrogen sulfite, one of the sulfites that can be used as a preservative, antioxidant and anti-browning agent in food with the European food additive number E222.
Does sodium bisulfite remove chlorine?
Sodium Bisulfite Sodium bisulfite reduces free chlorine to sodium bisulfate and hydrochloric acid. The chloramines are converted to sodium bisulfate, hydrochloric acid, and ammonium chloride. The theoretical dosage for dechlorination requires 1.78 parts of pure sodium sulfite per part chlorine or chloramines.
What is the pH of sodium bisulfite?
Sodium Bisulfite, wt%38.0 – 42.0pH3.6 – 4.6
How do you write sodium bisulfite?
Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) is a chemical mixture with the approximate chemical formula NaHSO3.
What is the function of sodium bisulfite?
Sodium bisulfite is a food additive, antioxidant, antibrowning and preservant. Upon heating in aqueous acidic media, it releases SO2. In dried fruits and jams, it acts as anti-browning agent by preventing enzymatic browning caused by polyphenol oxidase.
How do you use sodium bisulfite?
Sodium Bisulfite is also useful in stronger doses with water for sanitizing your equipment. The recommended dose is 1 teaspoon of Sodium Bisulfite per gallon of water. It is also recommended that either Citric Acid, Tartaric Acid or Acid Blend be added to the solution at the rate of 1/2 teaspoon per gallon.
Can I use sodium metabisulfite instead of sodium bisulfite?
sodium bisulfite is an old name for sodiumhydrogensulfite NaHSO3 and does not exist in solid form. In the solid form the formula is Na2S2O5 which is Sodium disulfite and this has the old name sodium metabisulfite. So if in your formula sodiumbisulfite (in solid form) is required, take the metabisulfite.
How do you make sodium bisulfite from sodium metabisulfite?
- Add 4.75 g of sodium metabisulfite (di-sodium disulfite, Na2S2O5) to 6.25 mL of sterile, degassed ddH2O.
- Add 1.75 mL of 2 m NaOH.
- Add 1.25 mL of 1 m hydroquinone (0.11 g in 1 mL of H2O).
- Heat to 50°C in the dark, inverting the tube frequently.
- Adjust the pH to 5.0.
How do you make sodium metabisulfite at home?
Sodium metabisulfite can be produced by crystallizing a solution of sodium bisulfite. Sodium bisulfite results from introducing sulfur dioxide into a solution of sodium sulfite. Sodium sulfite, in turn, can be produced by introducing sulfur dioxide into sodium hydroxide.
How do you quench sodium hypochlorite?
Sodium thiosulfate is a commonly used quenching agent for sodium hypochlorite and has previously been deemed acceptable when quenching hypochlorite solutions before conducting chlorate analysis.
Is sodium bisulfite a preservative?
Chemists make sodium bisulfite by combining sulfuric acid and table salt. Manufacturers use this substance as a food preservative, so you can find it listed on many food labels. Unfortunately, sodium bisulfite and a related compound — sodium metabisulfite — can cause serious side effects.
Is sodium bisulfate toxic?
Sodium bisulfate is a dry acid that may be harmful if swallowed in large amounts.
Why is sodium nitrite bad for you?
Sodium nitrate, a preservative that’s used in some processed meats, such as bacon, jerky and luncheon meats, could increase your heart disease risk. It’s thought that sodium nitrate may damage your blood vessels, making your arteries more likely to harden and narrow, leading to heart disease.