.
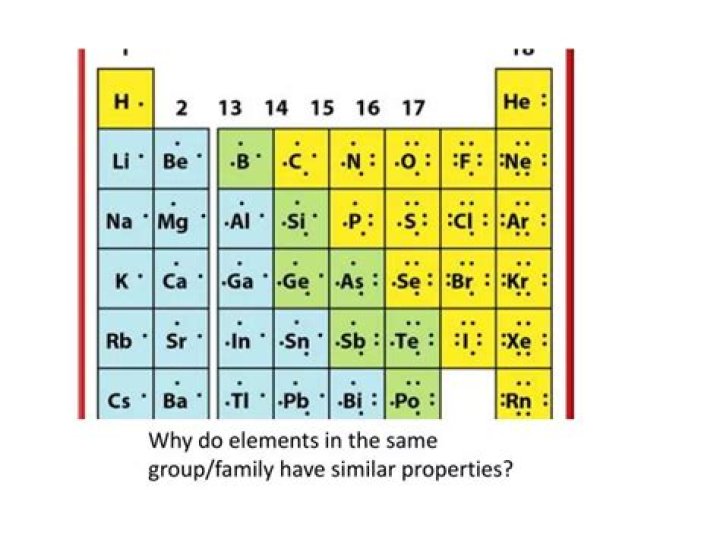
Then, why do elements in same group have similar properties?
Elements in the same group have similar properties because they have the same number of valence electrons in their outermost shell.
Likewise, what do elements in the same group have in common? The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. They are the electrons involved in chemical bonds with other elements. Every element in the first column (group one) has one electron in its outer shell.
Besides, do elements in the same group have the same physical properties?
Elements in the same group (in the periodic table) have similar chemical properties. This is because their atoms have the same number of electrons in the highest occupied energy level. The number of valence electrons in a particular group remains the same and this they have the same physical and chemical properties.
What similar properties do group 1 elements have?
Group one elements share common characteristics. They are all soft, silver metals. Due to their low ionization energy, these metals have low melting points and are highly reactive. The reactivity of this family increases as you move down the table.
Related Question AnswersWhich groups are the most reactive?
The two most reactive groups of elements are the alkali metals and the halogens, because of their valence electrons. Those groups have 1 and 7 valence electrons, respectively, making them desperate to bond to something, to achieve a stable configuration of 8.What group of elements are the most reactive nonmetals?
Answer and Explanation: The most reactive nonmetal is fluorine. Fluorine is a halogen, which is Group 17 on the periodic table, and the halogens are the most reactiveWhat determines the blocks in the periodic table?
A block of the periodic table is a set of chemical elements having their differentiating electrons predominately in the same type of atomic orbital. A differentiating electron is the electron that differentiates an element from the previous one.Why do we classify elements?
The number of elements discovered has increased, making it difficult to remember the behavior and properties of these elements. Hence it is important to classify elements according to their properties. It helps us understand how different elements form different compounds.What elements did Mendeleev predict?
Mendeleev's Predicted Elements- Eka-boron (scandium)
- Eka-aluminium (gallium)
- Eka-manganese (technetium)
- Eka-silicon (germanium)
What are the periodic trends in electronegativity?
Electronegativity increases as you move from left to right across a period on the periodic table. This is because, even though there are the same number of energy levels, there are more positive protons in the nucleus, creating a stronger pull on the negative electrons in the outer shell.Why do elements in a group show same Valency?
Elements in same group show same valency because they have same number of electrons in ther outermost shell i.e. Valence shell. Since valency depends upon the valence electrons so they have same valency. Elements in the same group have same valancy. These elements also have similar physical and chemical properties.Why are the elements in Group 0 unreactive?
The group 0 elements are placed in the rightmost column of the periodic table. They are called the noble gases because they are all chemically unreactive (the term noble in chemistry signifies a lack of chemical reactivity). They are unreactive because their atoms have stable arrangements of electrons.Why do elements in the same family have similar physical and chemical properties?
Elements in the same family have similar properties because they have the same number of valence electrons. What trend in atomic radius occurs down a group on the periodic table? Sodium and Potassium have the same number of valence electrons which control elements chemical properties.What kind of elements tend to gain electrons?
Elements that are metals tend to lose electrons and become positively charged ions called cations. Elements that are nonmetals tend to gain electrons and become negatively charged ions called anions. Metals that are located in column 1A of the periodic table form ions by losing one electron.Which family of elements have the highest melting points?
Other than carbon, tungsten (element 74, transition metal, relative atomic weight 183.84 ± 0.1, atomic symbol 'W') has the highest melting point at 3422°C. Tungsten also has the highest boiling point at a scorching 5555°C. Hope this helps!How can you determine if elements have the same chemical properties?
Two different elements have similar chemical properties when they have the same number of valence electrons in their outermost energy level. Elements in the same column of the Periodic Table have similar chemical properties. So what does that mean about their valence electrons?What are the three major categories of elements?
Of the three major categories of elements (metals, non-metals, and metalloids) which group is the largest?Why is argon unreactive?
Answer and Explanation: Argon is unreactive because it has the maximum number of electrons in its outer shell. According to the octet rule, most elements need to have 8Why do group 1 elements form positive ions?
Notice that all metals lose electrons to form positive ions. Group 1 metals commonly lose only one electron to form ions with a single positive charge. The nonmetals in Group 16 gain two electrons to form ions with a 2– charge. The noble gases do not normally gain or lose electrons and so do not normally form ions.Why do elements in groups share more chemical properties than elements in a period?
The elements of a group have similar chemical properties but in a period they have different chemical properties. This is because the chemicals properties depend on no of valence electrons.Why can there be no fixed position hydrogen?
The position of hydrogen is unsatisfactory in Mendeleev's periodic table. It was placed with Cu, Ag Au ,etc. Hydrogen is a metal which shows resemblance with halogenslike Cl, F, etc and with alkali metalslike Na, K, etc. That is why no fixed position was given to hydrogen in Mendeleev's periodic table.How do you know what group an element is in?
- For s-block elements , group number is equal to the number of valence electrons.
- For p-block elements ,group number is equal to 10+number of electrons electrons in the Valence shell .
- For d-block elements group number is equal to the number of electrons in a (n-1) d subshell + the number of electrons in Valence shell .